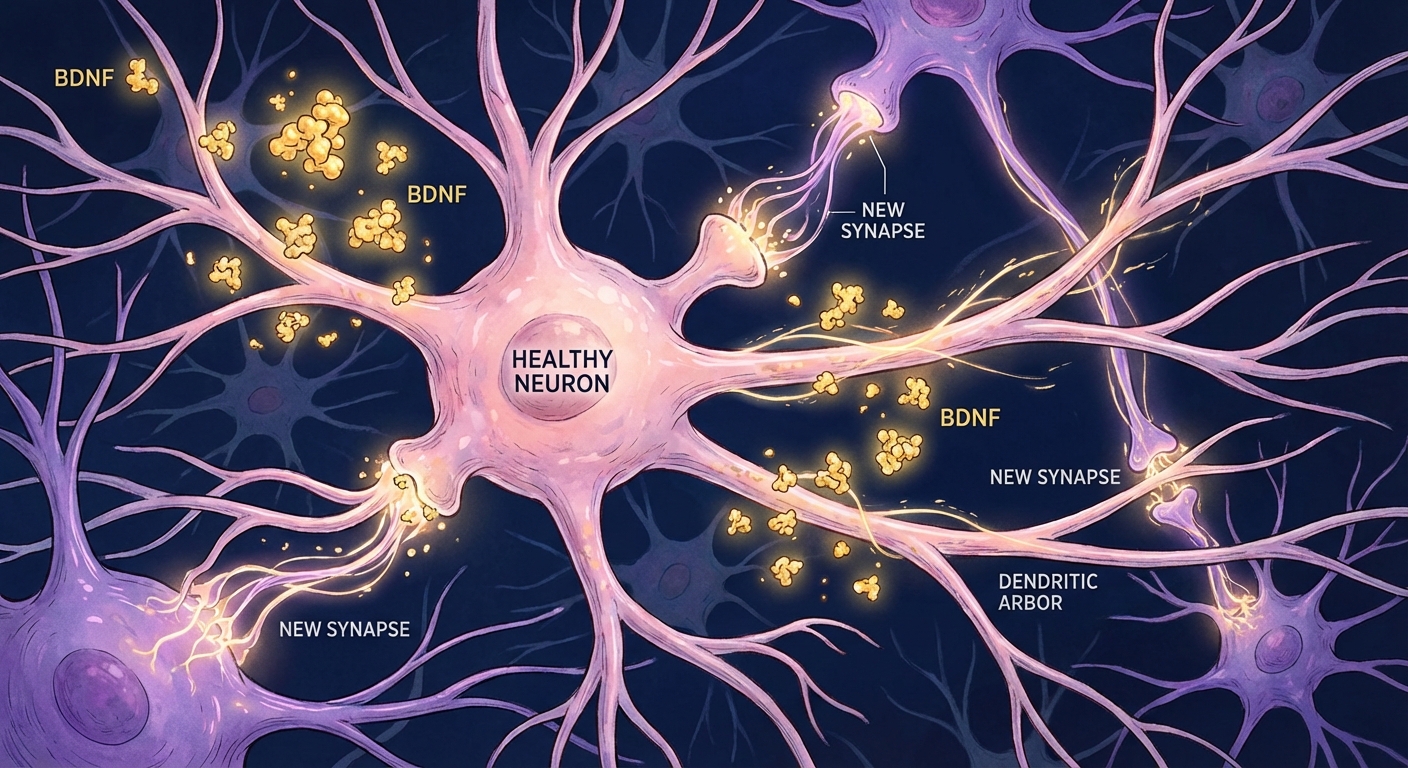
For decades, neuroscience taught that adult brains were essentially fixed, that the neurons you had at 25 were the neurons you’d die with, minus the inevitable decline. That dogma collapsed in the 1990s when researchers discovered that new neurons continue forming throughout life, particularly in the hippocampus where memory consolidation occurs. The protein primarily responsible for this ongoing brain renewal is BDNF, brain-derived neurotrophic factor, and understanding how to increase it has become one of the most actionable findings in modern neuroscience.
BDNF acts as fertilizer for your brain, supporting the survival of existing neurons while promoting the growth of new ones and the formation of synapses between them. Low BDNF levels correlate reliably with depression, cognitive decline, Alzheimer’s disease, and reduced learning capacity. High BDNF correlates with sharper cognition, better mood, larger hippocampal volume, and preserved brain function into old age. The critical finding that makes this clinically relevant: you can substantially increase your brain’s BDNF production through exercise, nutrition, sleep, and other lifestyle factors. Your BDNF levels aren’t fixed by genetics; they respond to how you live.
A single 30-minute session of vigorous exercise can increase BDNF by 30-50% for several hours afterward. Regular exercise sustained over months raises baseline BDNF levels by 20-50% even at rest. Specific foods including berries, fatty fish, and dark chocolate increase BDNF through distinct mechanisms. Sleep deprivation crashes BDNF while adequate sleep supports its production. This isn’t speculative future medicine, it’s actionable neuroscience you can implement today.
What BDNF Actually Does in Your Brain
BDNF belongs to a family of proteins called neurotrophins, molecules that support nerve cell survival and function. Among these, BDNF is the most abundant in the brain and the most responsive to lifestyle modification. Its effects span virtually every aspect of brain health that matters for cognitive function and mental wellbeing.
Neurogenesis, the production of new neurons, depends heavily on BDNF signaling. The hippocampus, your brain’s memory center, continues producing new neurons throughout life, a process that BDNF directly stimulates. Research from the Salk Institute published in Cell Stem Cell demonstrated that BDNF not only promotes the birth of new neurons but helps them integrate into existing neural circuits, a critical step for new neurons to become functional rather than dying off. Higher BDNF levels mean more new neurons survive and contribute to memory formation and learning.
Synaptic plasticity, the strengthening and weakening of connections between neurons that underlies all learning, requires BDNF for optimal function. When you learn something new, specific synapses strengthen through a process called long-term potentiation (LTP). BDNF is essential for this process, with neurons that lack BDNF showing dramatically impaired LTP in laboratory studies. Dr. Bai Lu at the National Institute of Child Health and Human Development has demonstrated that BDNF essentially acts as a “readiness signal” that primes synapses to encode new information. Without adequate BDNF, learning becomes slower and less durable.
Neuroprotection represents another critical BDNF function. The protein activates cellular defense mechanisms that protect neurons from oxidative stress, inflammation, and other damage that accumulates with aging. BDNF triggers the production of antioxidant enzymes and anti-inflammatory molecules within neurons themselves. This protective effect may explain why higher BDNF levels associate with slower brain aging and reduced dementia risk in longitudinal studies following participants over decades.
Mood regulation involves BDNF through its effects on serotonin and dopamine systems. The hippocampus and prefrontal cortex, brain regions affected by depression, show reduced BDNF levels in depressed patients compared to healthy controls. Successful antidepressant treatment, whether with medication, therapy, or exercise, increases BDNF as symptoms improve. This has led to the “neurotrophic hypothesis of depression,” which proposes that reduced neuroplasticity, mediated partly through low BDNF, contributes to depressive symptoms.
The Exercise-BDNF Connection
If you could only choose one intervention to increase BDNF, choose exercise. The research documenting exercise’s effects on BDNF is remarkably consistent across hundreds of studies spanning multiple decades, and the magnitude of effect rivals or exceeds any other intervention.
A single bout of aerobic exercise produces acute BDNF increases of 30-50% that persist for several hours post-exercise. A 2016 meta-analysis in the Journal of Psychiatric Research pooled data from 29 studies and found that a single exercise session reliably elevated peripheral BDNF levels, with effect sizes ranging from moderate to large depending on exercise intensity and duration. This immediate increase creates a window of enhanced neuroplasticity, which is why many researchers recommend learning or studying after exercise to capitalize on the elevated BDNF state.
Regular exercise sustained over weeks and months produces even more significant effects: chronically elevated baseline BDNF levels that persist throughout the day, not just after workouts. A 2020 systematic review in Neuroscience & Biobehavioral Reviews found that exercise training programs lasting 8+ weeks increased resting BDNF levels by 20-50% on average. The brain essentially resets its BDNF thermostat to a higher level with consistent training. This chronic elevation supports ongoing neuroplasticity, helping maintain cognitive function as you age.
Intensity matters. While any movement provides some benefit, higher-intensity exercise produces larger BDNF increases. A study comparing moderate continuous cycling to high-intensity intervals found that the interval group showed 30% greater BDNF elevation despite similar total work performed. This aligns with the concept of hormesis, where moderate stress triggers adaptive responses that strengthen the system. However, moderate intensity still produces meaningful effects, and the best exercise intensity is one you’ll actually do consistently.
Duration has a threshold effect. Studies suggest that 20-30 minutes represents a minimum effective dose for significant BDNF increase, with 30-60 minutes showing robust effects. Very short bouts (10-15 minutes) produce smaller, less consistent increases. For practical purposes, aiming for 30+ minutes of sustained cardiovascular activity appears optimal for BDNF specifically, though shorter sessions still provide other health benefits.

Nutrition Strategies That Increase BDNF
Diet influences BDNF through multiple pathways including direct effects of specific compounds, anti-inflammatory mechanisms, and gut-brain signaling. While no food intervention rivals exercise’s BDNF-boosting power, several nutritional approaches show meaningful effects and complement exercise’s benefits.
Omega-3 fatty acids, particularly DHA (docosahexaenoic acid), increase BDNF levels in both animal and human studies. DHA comprises approximately 15% of brain fatty acid content and serves as a structural component of neuronal membranes. A 2024 study in Nutritional Neuroscience found that 12 weeks of omega-3 supplementation (2g daily of combined EPA and DHA) increased BDNF levels by 15% in healthy adults compared to placebo. The effect was more pronounced in participants with lower baseline omega-3 status. Food sources include fatty fish (salmon, mackerel, sardines, herring), with 2-3 servings weekly providing adequate omega-3 for most people. For those who don’t eat fish, algae-derived DHA supplements provide a direct source.
Polyphenols and flavonoids activate molecular pathways that increase BDNF gene expression. Blueberries have been particularly well-studied, with anthocyanins (the compounds providing their deep color) shown to increase hippocampal BDNF and improve memory in randomized controlled trials. A 2023 study published in Frontiers in Nutrition found that 12 weeks of daily blueberry consumption improved episodic memory in older adults, with BDNF increases correlating with cognitive improvements. Dark chocolate provides flavanols that similarly increase BDNF, with benefits appearing at doses as low as 1-2 ounces daily of 70%+ cacao chocolate. Green tea’s EGCG (epigallocatechin gallate) shows BDNF-increasing effects in multiple studies, adding brain benefits to its metabolic and cardiovascular effects.
Intermittent fasting and time-restricted eating increase BDNF through metabolic stress pathways. When glucose availability drops during fasting periods, cells activate adaptive responses including increased BDNF production. Research from the National Institute on Aging found that alternate-day fasting in rodents increased hippocampal BDNF by 50-100%, with human studies showing more modest but still meaningful effects. A 16:8 eating pattern (eating within an 8-hour window daily) is the most studied and practical approach, producing BDNF increases without the difficulty of longer fasts.
Curcumin, the active compound in turmeric, increases BDNF in multiple studies, though bioavailability is a challenge. Standard turmeric powder provides minimal curcumin absorption. Consuming curcumin with black pepper (which contains piperine) increases absorption by 2,000%, or specialized formulations with phospholipids or nanoparticles can enhance bioavailability. A 2020 meta-analysis found that curcumin supplementation at 500-1,500mg daily increased BDNF in healthy adults, with effects more pronounced in older populations.
Lifestyle Factors Beyond Diet and Exercise
BDNF regulation extends beyond physical activity and nutrition to encompass sleep, stress, social connection, and cognitive engagement. Understanding these factors reveals how lifestyle choices compound to either support or undermine brain health.
Sleep is when significant BDNF-mediated brain maintenance occurs. BDNF levels rise during sleep, particularly during slow-wave deep sleep when memory consolidation happens. Sleep deprivation dramatically reduces BDNF: even a single night of poor sleep causes measurable BDNF decreases, while chronic sleep restriction produces sustained suppression. A study in Sleep journal found that sleeping less than 6 hours nightly for one week reduced BDNF by 40% compared to 8-hour sleepers. The relationship is bidirectional, as BDNF also promotes healthy sleep architecture, meaning poor sleep and low BDNF can create a vicious cycle. Prioritizing 7-9 hours of quality sleep supports BDNF production while allowing the brain restoration processes that BDNF facilitates.
Chronic stress suppresses BDNF through cortisol and inflammatory pathways. The hippocampus, the brain region most dependent on BDNF for ongoing function, is also highly sensitive to stress hormones. Chronic elevation of cortisol literally shrinks the hippocampus over time, an effect mediated partly through reduced BDNF. Stress management practices including meditation, yoga, time in nature, and social support all help maintain BDNF levels by reducing chronic stress activation. Sleep optimization represents one of the most powerful stress management tools, as quality sleep both reduces cortisol and directly supports BDNF production.

Social connection increases BDNF through mechanisms that aren’t fully understood but appear robust across studies. Meaningful social interaction, feelings of belonging, and positive relationships correlate with higher BDNF levels, while loneliness and social isolation associate with reduced BDNF. The effect may involve reduced stress hormone exposure, direct neurochemical effects of social bonding, or the cognitive stimulation that social interaction provides. Regardless of mechanism, maintaining strong social connections appears to be neuroprotective.
Learning and cognitive challenge stimulate BDNF production as the brain upregulates growth factors to support new neural connections. Novel learning, whether acquiring a new skill, studying a language, playing a musical instrument, or engaging in complex problem-solving, signals the brain to produce the molecular support structures needed for encoding new information. This is “use it or lose it” operating at the molecular level. Regular cognitive challenge maintains the neuroplasticity capacity that might otherwise decline with age.
The Depression and Dementia Connections
BDNF research has particular clinical relevance for two major brain health concerns: depression and cognitive decline. In both conditions, low BDNF appears to be both a biomarker and potentially a causal factor.
Depression shows one of the most consistent BDNF associations in psychiatry. Depressed patients have lower serum BDNF than healthy controls, with multiple meta-analyses confirming this relationship. Successful treatment, whether with antidepressant medications, psychotherapy, or exercise, increases BDNF as symptoms improve. The neurotrophic hypothesis of depression proposes that reduced neuroplasticity, mediated through low BDNF, contributes to the cognitive symptoms of depression (poor concentration, memory problems) and may underlie the condition itself. This hypothesis explains why antidepressants take weeks to work despite immediately affecting neurotransmitter levels: the therapeutic effect requires time to increase BDNF and restore neuroplasticity.
Exercise’s antidepressant effects, comparable to medication for mild-moderate depression in randomized trials, likely work substantially through BDNF. A 2023 study in JAMA Psychiatry found that exercise interventions produced larger BDNF increases than medication alone, with BDNF changes correlating with symptom improvement. This provides a neurobiological rationale for exercise as a depression treatment and prevention strategy, not just a complementary approach but a primary intervention targeting the same molecular pathways as pharmaceutical treatments.
Dementia prevention involves BDNF through multiple mechanisms. Lower midlife BDNF levels predict higher dementia risk decades later in longitudinal studies following participants for 20-30 years. Higher BDNF correlates with larger hippocampal volume, better memory performance, and reduced Alzheimer’s pathology in post-mortem studies. The relationship is strong enough that some researchers have proposed BDNF as a biomarker for brain aging and dementia risk.
Interventions that increase BDNF, including exercise, omega-3 consumption, polyphenol-rich diets, and ongoing cognitive engagement, independently reduce dementia risk in separate research. While causality isn’t definitively proven, the convergence of evidence strongly suggests that lifestyle factors supporting BDNF also protect long-term brain health. The practical implication: investing in BDNF-supporting habits in midlife may pay dividends in preserved cognitive function decades later.
A Practical BDNF-Boosting Protocol
Translating BDNF research into daily practice involves combining multiple interventions that work through complementary mechanisms. No single approach maximizes BDNF; the most powerful strategy integrates exercise, nutrition, and lifestyle factors.
Daily foundations should include 30-60 minutes of aerobic exercise, the single most powerful BDNF intervention. Running, cycling, swimming, or any sustained cardiovascular activity that elevates heart rate provides robust BDNF increases. Consume polyphenol-rich foods including 1-2 cups of berries, green tea, or 1-2 ounces of dark chocolate. Ensure omega-3 intake through fatty fish 2-3 times weekly or 1-2g of EPA/DHA from supplements. Sleep 7-9 hours with attention to sleep quality, not just duration.
Weekly additions include 2-3 resistance training sessions that complement aerobic exercise’s brain benefits. Engage in novel learning or cognitively challenging activities, whether studying something new, practicing an instrument, or engaging in complex problem-solving. Maintain meaningful social connections through regular interaction with people who matter to you.
Practices to minimize include chronic unmanaged stress (address through meditation, yoga, therapy, or tackling root causes), sleep deprivation (one of the fastest ways to crash BDNF), ultra-processed foods and excessive sugar (which promote inflammation and impair brain function), and social isolation (which reduces BDNF through mechanisms we’re still understanding).
Optional supplements with BDNF research support include lion’s mane mushroom (500-1,000mg daily, the supplement with strongest BDNF evidence), omega-3 if not getting adequate fish intake, and curcumin in bioavailable form (500-1,500mg with piperine or specialized formulation). These supplement the foundation of exercise and nutrition rather than replacing them.
The Bottom Line
BDNF represents one critical pathway through which lifestyle affects brain health, translating your daily choices about exercise, food, sleep, and stress into molecular signals that maintain or undermine cognitive function. Higher BDNF promotes neurogenesis, strengthens synapses, protects neurons from damage, and supports the mood regulation systems affected in depression. Lower BDNF correlates with cognitive decline, depression, and accelerated brain aging.
The actionable finding is that BDNF responds robustly to lifestyle modification. Exercise remains the most powerful intervention, with a single session producing 30-50% increases and regular training elevating baseline levels by 20-50%. Nutrition provides complementary effects through omega-3s, polyphenols, and strategic fasting. Sleep, stress management, social connection, and cognitive engagement round out a comprehensive approach to BDNF optimization.
Your brain’s capacity for learning, memory, and resilience isn’t fixed at birth or locked in by your twenties. It responds to how you live, day by day and year by year. The choices you make about movement, food, sleep, and connection accumulate into the brain function you’ll have at 60, 70, and beyond. The gut-brain axis adds another dimension to this picture, as intestinal health influences both inflammation and neurotransmitter production in ways that affect BDNF signaling. Start investing in your brain’s future now, while prevention and optimization remain possible, and let BDNF be the fertilizer that keeps your neural garden growing.
Sources: Cell Stem Cell (Salk Institute neurogenesis research), Journal of Psychiatric Research (exercise-BDNF meta-analysis), Neuroscience & Biobehavioral Reviews (training effects), Nutritional Neuroscience (omega-3 studies), Frontiers in Nutrition (blueberry research), National Institute on Aging (fasting research), Sleep journal (sleep deprivation effects), JAMA Psychiatry (exercise and depression).