Your father keeps asking people to repeat themselves. He turns the television up louder each month. At family dinners, he’s started nodding along to conversations you suspect he can’t fully hear. You’ve mentioned hearing aids, and he’s waved you off every time. A new seven-year study tracking nearly 2,800 older adults may give you the most persuasive argument yet: wearing hearing aids was associated with a 33% reduction in the risk of developing dementia, even though standard cognitive tests couldn’t detect measurable improvements in memory or thinking scores.
The research, led by Joanne Ryan, PhD, at Monash University in Melbourne and published in the journal Neurology in February 2026, represents one of the longest and largest prospective studies to examine the hearing aid-dementia connection. Its findings arrive at a critical moment, as the 2024 Lancet Commission identified hearing loss as the single largest modifiable risk factor for dementia, accounting for roughly 7% of all dementia cases worldwide. That percentage may sound small until you consider it translates to millions of potentially preventable diagnoses.
Inside the Study: Seven Years of Tracking
The research team followed 2,777 Australian adults with an average age of approximately 75, all of whom reported moderate hearing loss and none of whom had dementia at baseline. Of this group, 664 participants received hearing aid prescriptions during the study period, while the remainder served as the comparison group. Researchers tracked all participants annually with standardized cognitive assessments measuring memory, language, and processing speed over the full seven-year follow-up.
The headline finding was both striking and paradoxical. Participants who were prescribed hearing aids showed no significant improvement on standardized cognitive test scores compared to those who weren’t prescribed hearing aids. By conventional measures of memory and thinking ability, the two groups looked essentially identical throughout the study.
But when researchers looked at actual dementia diagnoses rather than test scores, a dramatically different picture emerged. Just 5% of participants prescribed hearing aids developed dementia during the study, compared to 8% of those without hearing aids. After adjusting for confounding factors including age, sex, diabetes, and heart disease, this translated to a 33% lower risk of dementia in the hearing aid group. The protective pattern extended to broader cognitive impairment as well: 36% of the hearing aid group developed any form of cognitive impairment versus 42% in the comparison group, representing a 15% lower risk.
Perhaps most compellingly, the study found a dose-response relationship. More consistent hearing aid use correlated with progressively lower dementia risk, suggesting that the protective effect wasn’t simply a matter of having hearing aids available but of actually wearing them regularly.
The Paradox: Protection Without Measurable Cognitive Improvement
The study’s most intellectually interesting finding is the disconnect between cognitive test performance and dementia diagnosis. How can hearing aids reduce dementia risk by a third while producing no detectable improvement on memory and thinking tests?
Dr. Ryan and her colleagues proposed several explanations. Standard cognitive assessments measure specific domains like working memory, verbal fluency, and processing speed at a single point in time. They’re designed to detect relatively large changes over short intervals. Dementia, by contrast, develops through gradual neurological changes that accumulate over years or decades before crossing the threshold into clinical diagnosis. Hearing aids may be slowing this accumulation in ways that don’t register on annual tests but meaningfully delay the point at which pathology becomes diagnosable disease.
There’s also the measurement sensitivity problem. Most cognitive tests weren’t designed for people with hearing loss. If a participant can’t hear the instructions clearly, their “cognitive” score may actually reflect their hearing deficit rather than their thinking ability. This means the tests may be too blunt an instrument to detect the specific type of cognitive preservation that hearing aids provide.
The research suggests that hearing aids may protect the brain through mechanisms that operate below the resolution of conventional testing, quietly preserving neural networks and social connections that collectively delay the clinical expression of dementia. This is consistent with the concept of “cognitive reserve,” the brain’s ability to tolerate pathological damage before symptoms appear, and suggests that maintaining auditory input may build or preserve this reserve in ways we’re only beginning to measure.
Four Theories on How Hearing Loss Drives Cognitive Decline
The biological plausibility of the hearing aid-dementia connection rests on several well-established mechanisms, each supported by independent lines of evidence. Understanding these pathways helps explain why correcting hearing loss could have such a profound impact on brain health.
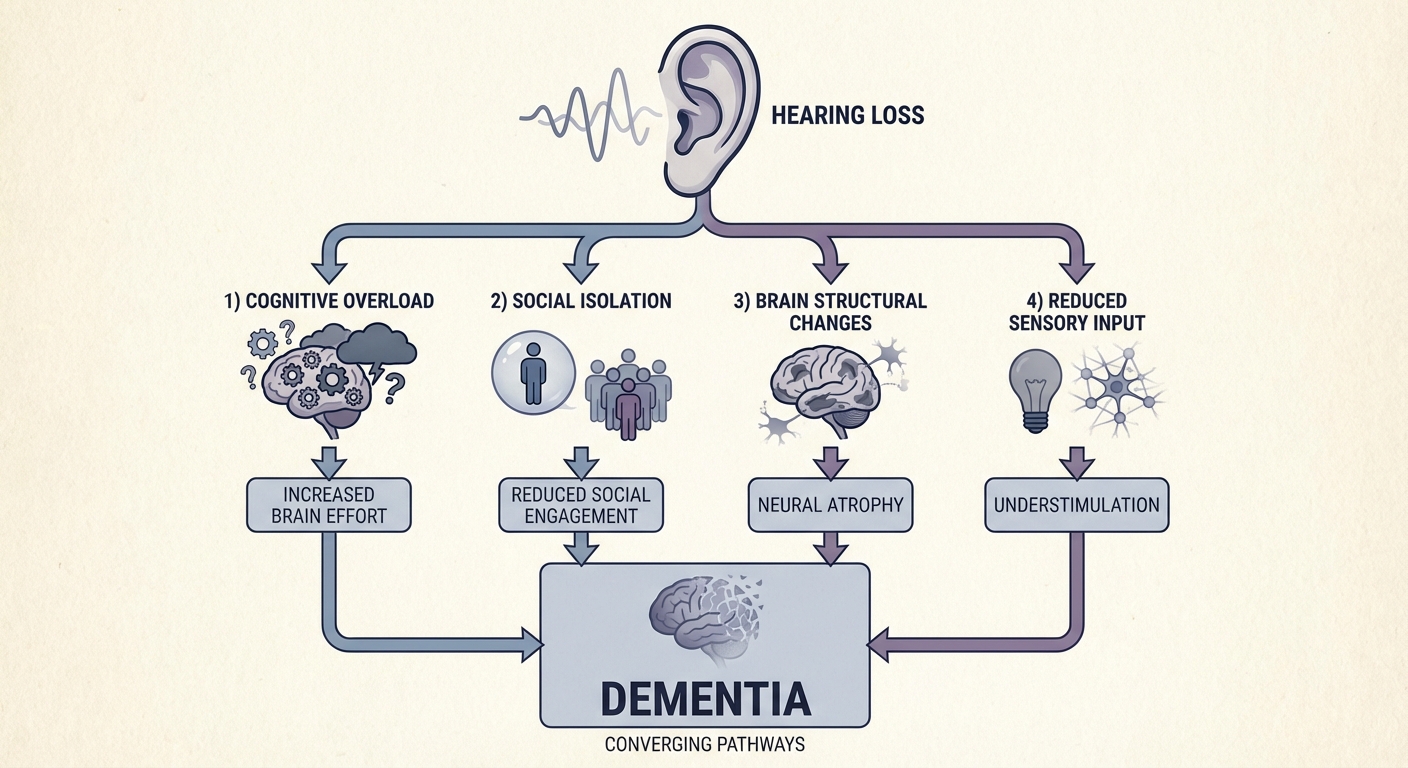
The cognitive load hypothesis posits that when auditory input degrades, the brain must allocate disproportionate resources to the basic task of hearing. Studies using pupillometry (which measures cognitive effort through pupil dilation) have confirmed that people with hearing loss show significantly greater physiological effort during listening tasks. This constant reallocation of cognitive resources may leave fewer neural resources available for memory encoding, executive function, and other higher-order processes. Over years, this chronic overallocation could accelerate cognitive decline.
The sensory deprivation theory focuses on structural brain changes. Prolonged hearing loss has been associated with reduced volume in the temporal lobes, the brain regions responsible for processing sound but also critically involved in memory and language. A 2023 study using high-resolution MRI found that adults with untreated moderate hearing loss showed measurable temporal lobe atrophy compared to age-matched controls with normal hearing or treated hearing loss. The brain, following the “use it or lose it” principle, appears to physically remodel in response to reduced auditory stimulation.
The information degradation model suggests that compromised auditory signals force the brain to process lower-quality information, leading to downstream errors in comprehension, memory formation, and recall. Think of it like trying to read a book with every third word blurred out. You can still follow the general narrative, but the effort required increases dramatically, and your comprehension of subtle details suffers. Over time, this reduced information quality may compound into measurable cognitive deficits.
Finally, the social isolation pathway may be the most straightforward. Hearing loss makes conversation difficult, embarrassing, and exhausting. People with untreated hearing loss progressively withdraw from social situations, family gatherings, and community activities. Social isolation is itself a well-established independent risk factor for dementia, with some studies suggesting it increases dementia risk by 50-60%. By restoring the ability to participate in conversation, hearing aids may protect cognition primarily by preserving social engagement.
The reality is that all four mechanisms likely operate simultaneously, creating a compounding effect. This convergence helps explain why hearing correction can produce such a disproportionately large reduction in dementia risk relative to what any single mechanism would predict.
Putting This in Context: The Broader Evidence
The Monash study doesn’t exist in isolation. It builds on a growing body of research connecting hearing intervention to cognitive protection. The landmark ACHIEVE trial, conducted across multiple U.S. sites, found that hearing aid use over three years cut cognitive decline in half among older adults already at elevated risk for dementia. The National Institutes of Health highlighted these findings as evidence that hearing intervention represents one of the most actionable dementia prevention strategies available.
The 2024 Lancet Commission on dementia prevention, intervention, and care identified hearing loss as the largest single modifiable risk factor for dementia, accounting for approximately 7% of all cases. To put that in perspective, physical inactivity accounts for roughly 2%, depression about 3%, and social isolation around 4%. Hearing loss surpasses all of them individually. The Commission estimated that eliminating hearing loss as a risk factor could potentially prevent or delay up to 800,000 dementia cases annually worldwide.
However, not all researchers agree on the strength of the causal claim. A February 2026 review published in Frontiers in Dementia cautioned that hearing devices “should not be marketed for dementia risk reduction,” noting that the evidence, while suggestive, hasn’t definitively established causation. The review emphasized that hearing aids should be recommended for their quality-of-life benefits, better communication, and social participation, rather than being positioned primarily as a dementia prevention tool. This is an important distinction, because overstating the cognitive benefits could paradoxically increase stigma around hearing loss by implying that people who don’t treat it are “choosing” dementia.
For those tracking the broader landscape of brain health and cognitive wellness, the hearing aid evidence adds another piece to the prevention puzzle. Combined with research on circadian rhythm disruption and dementia risk, a picture is emerging where dementia prevention is less about any single intervention and more about addressing multiple modifiable risk factors simultaneously.
The Treatment Gap: Who Needs Hearing Aids and Who Actually Gets Them
The practical challenge isn’t the science. It’s adoption. Among adults aged 70 and older with hearing loss that could benefit from hearing aids, only about 30% actually use them. In adults aged 20 to 69, that number drops below 16%. The reasons are well-documented and stubbornly persistent: cost (hearing aids can run $2,000 to $7,000 per pair), stigma (association with aging and disability), discomfort (adjustment period and maintenance requirements), and denial (gradual hearing loss is easy to rationalize as “everyone mumbles these days”).

The landscape has shifted somewhat since the FDA’s 2022 ruling allowing over-the-counter hearing aids for mild to moderate hearing loss, which reduced entry-level prices to the $200 to $1,000 range. Modern devices are also dramatically smaller, more comfortable, and more technologically sophisticated than even five years ago, with Bluetooth connectivity, smartphone app control, and adaptive noise processing that would have been science fiction a generation ago.
The Monash study adds urgency to the adoption conversation. If hearing aids can reduce dementia risk by a third, and if the dose-response relationship holds (more consistent use equals greater protection), then the years spent in denial about hearing loss aren’t just years of missed conversations. They may be years of accelerating neurological vulnerability that hearing correction could have slowed or prevented.
For anyone noticing signs of hearing difficulty, whether in yourself or a family member, the evidence now supports a proactive rather than reactive approach. The earlier hearing loss is addressed, the more years of potential cognitive protection hearing aids may provide. Given the study’s finding that consistent use matters more than simply owning the devices, the goal isn’t just to get hearing aids but to wear them daily and in a wide variety of listening environments.
What the Evidence Tells Us
The Monash University study adds the strongest longitudinal evidence yet to the case for hearing intervention as a cognitive health strategy. A 33% reduction in dementia risk over seven years, with a dose-response relationship favoring consistent use, represents a meaningful and actionable finding for the estimated 1.5 billion people worldwide living with some degree of hearing loss.
Three steps worth considering. First, if you’re over 50, get a baseline hearing assessment even if you don’t think you have a problem, because hearing loss is typically gradual and easy to miss until it’s moderate or severe. Second, if hearing loss is detected, explore treatment promptly rather than waiting years while the cognitive load, social isolation, and sensory deprivation mechanisms may already be operating. Third, if you or a family member already have hearing aids, prioritize consistent daily use, since the study suggests that protection scales with how reliably the devices are worn.
The research doesn’t claim that hearing aids prevent dementia outright. What it does suggest, with seven years of data from nearly 2,800 participants, is that treating hearing loss may be one of the most accessible and cost-effective tools we have for reducing dementia risk. In a disease with no cure and limited treatment options, a 33% risk reduction from a well-established, widely available medical device is not a finding to ignore. For the health technology landscape, hearing aids may be the most underrated longevity tool hiding in plain sight.
Sources
- Hearing aids didn’t boost memory tests but dementia risk dropped - ScienceDaily, February 2026
- Treating Hearing Loss With Hearing Aids for the Prevention of Cognitive Decline and Dementia - Neurology, February 2026
- Hearing aids slow cognitive decline in people at high risk - National Institutes of Health
- Dementia and hearing loss: from risk to mechanisms and management - Frontiers in Dementia, 2026