You’ve been told to take fish oil. Your doctor recommended it. The bottle on your counter promises heart health, brain health, and anti-inflammatory benefits. But what if the same supplement that protects one person from colorectal cancer actually accelerates tumor growth in another?
That’s the unsettling finding from a study published in Cellular and Molecular Gastroenterology and Hepatology by researchers at the University of Michigan and the University of Texas MD Anderson Cancer Center. The research identifies a single enzyme, 15-lipoxygenase-1 (encoded by the ALOX15 gene), as the gatekeeper that determines whether EPA and DHA, the two primary omega-3 fatty acids in fish oil, suppress colorectal tumors or potentially promote them. The implications challenge the assumption that fish oil is universally beneficial, and they suggest that personalized genetic screening may eventually become part of cancer prevention protocols.
This isn’t a minor footnote in the supplement literature. It’s a fundamental rethinking of how omega-3 fatty acids interact with individual biology, and it arrives at a time when roughly 18 million Americans take fish oil daily, many of them specifically hoping to reduce cancer risk.
The ALOX15 Gatekeeper: How One Enzyme Changes Everything
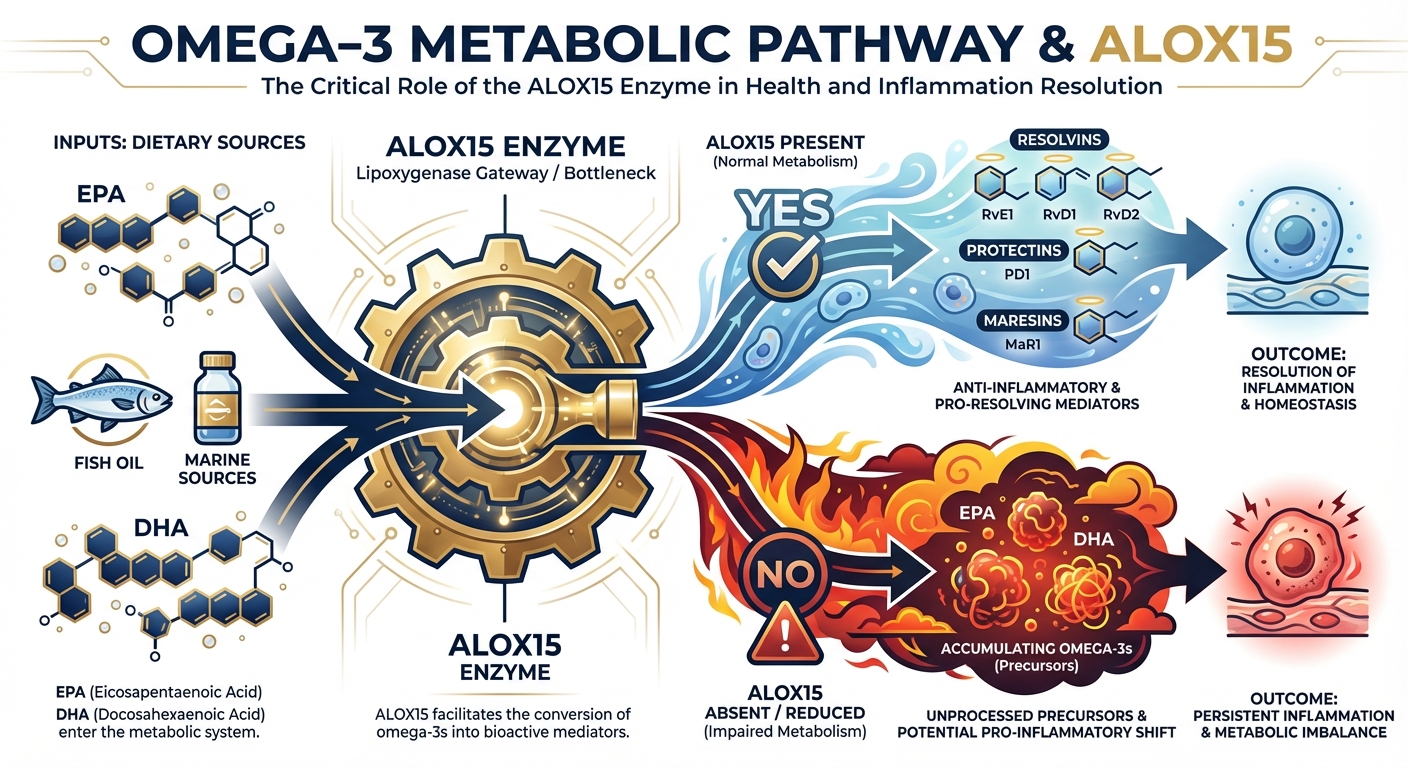
To understand why fish oil can have opposite effects in different people, you need to understand the resolvin pathway. When you consume EPA and DHA from fish oil, your body doesn’t use them directly as anti-inflammatory agents. Instead, they serve as raw materials that must be converted into specialized pro-resolving mediators (SPMs), including resolvins, protectins, and maresins. These SPMs are the actual molecular workhorses that reduce chronic inflammation and suppress tumor-promoting environments in the gut.
The enzyme ALOX15 is the critical bottleneck in this conversion process. It catalyzes the first step of converting EPA into resolvin E1 and DHA into resolvin D1, the compounds that actively resolve inflammation rather than simply suppressing it. Without functional ALOX15, the conversion stalls. The omega-3 fatty acids accumulate without being processed into their protective forms, and the excess appears to create conditions that may actually promote tumor development.
“Not all fish oil supplements are the same,” said Imad Shureiqi, professor of internal medicine at the University of Michigan and a member of the Rogel Cancer Center. “It is also important to ask whether the person who is taking the supplement has the required enzymes to metabolize these products to prevent chronic inflammation and subsequently cancer development.”
This distinction matters because ALOX15 expression varies significantly between individuals and is frequently silenced in colorectal cancer tissue. Research published in Cancer Research has shown that ALOX15 is downregulated in roughly 60-70% of colorectal tumors, meaning the majority of people who already have colorectal cancer may lack the enzyme needed to benefit from fish oil supplementation.
EPA vs. DHA: Not All Omega-3s Are Equal
One of the study’s most striking findings is that EPA and DHA behave very differently when ALOX15 is absent. In mice with functional ALOX15, both fatty acids showed tumor-suppressive effects, though EPA consistently outperformed DHA. The ethyl ester and free fatty acid forms of EPA reduced both tumor number and tumor volume in mice with active ALOX15, and Lovaza (a prescription omega-3 ethyl ester) showed particularly strong results.
But when ALOX15 was knocked out, the two fatty acids diverged dramatically. EPA-fed mice without ALOX15 showed modest increases in tumor burden, but DHA-fed mice without the enzyme showed significantly more tumors. The researchers observed that DHA variants failed to prevent tumor growth in ALOX15-deficient mice and, in some experimental conditions, appeared to actively promote tumorigenesis.
This differential response likely stems from the distinct metabolic fates of EPA and DHA. EPA can be partially processed through alternative enzymatic pathways (including COX-2 and CYP450 enzymes) to produce some anti-inflammatory mediators even without ALOX15. DHA, by contrast, is more dependent on the ALOX15 pathway for conversion to its protective resolvin D series. When that pathway is blocked, DHA may be metabolized through pro-inflammatory routes instead, generating compounds that promote rather than suppress tumor growth.

The practical implication is significant. If future research confirms these findings in humans, people with low ALOX15 expression might benefit more from EPA-dominant supplements than from balanced EPA/DHA formulas or DHA-heavy products. Currently, most fish oil supplements contain both fatty acids in varying ratios, and the distinction is rarely discussed in clinical recommendations.
The Personalized Prevention Question: Should You Test for ALOX15?
Here’s where this research gets genuinely provocative: if ALOX15 status determines whether fish oil helps or harms cancer risk, should genetic or enzymatic screening become standard before recommending omega-3 supplements?
The answer, right now, is “not yet, but possibly soon.” ALOX15 expression isn’t a simple on/off genetic switch. The gene can be present in your DNA but silenced through epigenetic mechanisms, particularly DNA methylation. This means a standard genetic test wouldn’t capture the full picture. You’d need tissue-level expression data, which currently requires a biopsy, to know whether your colorectal tissue is actually producing ALOX15. That’s not a practical screening tool for the general population.
However, the Shureiqi lab at Michigan is already developing pharmacological approaches to reactivate ALOX15 in cancer cells. If successful, these drugs could serve dual purposes: restoring the ability of omega-3 supplements to produce anti-inflammatory resolvins in cancer tissue, while simultaneously removing a tumor-promoting pathway. The combination of ALOX15 reactivation with targeted EPA supplementation represents a genuinely novel approach to colorectal cancer prevention.
There are also simpler screening possibilities on the horizon. Liquid biopsy technology, which detects tumor-derived molecules in blood samples, is advancing rapidly. Future tests might measure circulating resolvin levels as a proxy for ALOX15 activity. If your blood shows adequate resolvin production after fish oil supplementation, your ALOX15 pathway is likely functional. If resolvin levels don’t respond to omega-3 intake, you may lack the enzymatic machinery to benefit from supplementation.
What We Already Knew, and What This Changes
The relationship between omega-3s and cancer has been debated for decades. Previous research has been frustratingly inconsistent: some large-scale studies show protective effects, others show no benefit, and a few suggest potential harm. The 2019 VITAL trial, which randomized 25,871 adults to receive omega-3 supplements or placebo, found no overall reduction in cancer incidence. But subgroup analyses have consistently hinted that certain populations respond differently.
This ALOX15 research offers a biological explanation for that inconsistency. If roughly 60-70% of colorectal tumors have silenced ALOX15, and if a meaningful percentage of the general population has reduced ALOX15 expression in their gut tissue, then clinical trials averaging across all participants would naturally dilute any protective signal. The responders and non-responders cancel each other out in aggregate data.

This is a pattern we’ve seen before in nutrition research. The failure to account for individual biological variation has undermined countless supplement trials. Vitamin D supplementation trials, for example, showed inconsistent results for years until researchers began stratifying by baseline vitamin D status and discovered that benefits concentrated in deficient populations. The same principle may apply to fish oil: the supplement works when the biological machinery to process it is present, and it doesn’t (or may even backfire) when that machinery is absent.
The ALOX15 finding also reshapes how we think about the broader omega-3 deficiency crisis. If a significant portion of the population cannot effectively convert omega-3s into resolvins, simply increasing intake won’t solve the problem. The focus may need to shift from “how much omega-3 should you consume?” to “can your body actually use the omega-3 you’re consuming?”
A Framework for Thinking About Conditional Supplementation
This study illuminates a broader principle that extends well beyond fish oil: the era of universal supplement recommendations may be ending. We’re entering what I’d call “conditional supplementation,” where the question isn’t just “what should I take?” but “does my biology support taking this?”
Consider the parallels. Folate supplementation is beneficial for most people but can accelerate tumor growth in individuals with existing colorectal polyps. Calcium supplementation strengthens bones in most populations but may increase cardiovascular risk in certain subgroups. Beta-carotene supplements were once widely recommended until trials showed they increased lung cancer risk specifically in smokers. In each case, a supplement that’s helpful for one population becomes harmful for another, and the distinguishing factor is individual biology.
The ALOX15 story fits this pattern perfectly. Fish oil isn’t inherently good or bad for cancer prevention. It’s conditionally beneficial, depending on whether your body has the enzymatic tools to convert it into protective compounds. This framing doesn’t mean you should stop taking fish oil. It means the blanket recommendation “everyone should take omega-3s” deserves more nuance than it typically receives. For individuals at elevated colorectal cancer risk, this nuance could be medically significant.
The Verdict on Fish Oil
If you’re currently taking fish oil for general health, this research shouldn’t trigger panic. The cardiovascular and neurological benefits of omega-3 supplementation operate through pathways beyond ALOX15, and the cancer-specific findings are currently limited to mouse models. Human trials will be needed to confirm whether ALOX15 status predicts fish oil response in people.
That said, several practical considerations emerge from this research.
For general health: Continue omega-3 supplementation if your physician recommends it, particularly for cardiovascular health. The anti-inflammatory benefits through non-ALOX15 pathways are well-established in human trials.
For cancer risk reduction: If you have a personal or family history of colorectal cancer, discuss this research with your oncologist. EPA-dominant formulations may be preferable to high-DHA products based on the differential findings in this study.
For the supplement industry: This research underscores the need for more precise labeling and consumer education. The EPA-to-DHA ratio in fish oil supplements varies widely, from 2:1 EPA-dominant to 1:2 DHA-dominant, and most consumers have no basis for choosing between them.
For future research: The Shureiqi lab’s work on ALOX15 reactivation drugs could open an entirely new category of combination therapy, pairing dietary supplements with pharmacological agents that restore the body’s ability to use them. Watch for clinical trials in the next 2-3 years.
The fish oil question isn’t “should you take it?” anymore. It’s becoming “can your body use it?” That’s a fundamentally different question, and answering it could transform how we approach personalized cancer prevention.
Sources
- Zuo X, Kiyasu Y, Liu Y, et al. “Colorectal 15-Lipoxygenase-1 as a Host Factor Determining the Effects of Eicosapentaenoic Acid and Docosahexaenoic Acid on Colorectal Tumorigenesis in Mice.” Cellular and Molecular Gastroenterology and Hepatology, 2025. https://www.cmghjournal.org/article/S2352-345X(25)00148-1/fulltext
- Michigan Medicine Health Lab. “Fish Oil Supplements May Not Work for Certain Cancer Patients.” University of Michigan, 2026. https://www.technologynetworks.com/genomics/news/gene-found-to-determine-whether-fish-oil-protects-against-cancer-405147
- Manson JE, et al. “Marine n-3 Fatty Acids and Prevention of Cardiovascular Disease and Cancer.” New England Journal of Medicine, 2019. VITAL Trial.
- Shureiqi I, et al. “ALOX15 as a Suppressor of Inflammation and Cancer: Lost in the Link.” Prostaglandins and Other Lipid Mediators, 2017. https://pmc.ncbi.nlm.nih.gov/articles/PMC5509529/