The longevity industry has operated under a reassuring assumption for the past two decades: your genes load the gun, but your lifestyle pulls the trigger. The implication is empowering. Eat right, exercise, sleep well, manage stress, and you can outrun whatever genetic hand you were dealt. Supplement companies, biohacking gurus, and longevity clinics have all built their pitch on this premise, often citing a widely repeated statistic that genetics account for only 20 to 25% of lifespan variation. Some estimates went even lower. A 2018 study from Calico Life Sciences, the longevity arm of Alphabet, pegged the number below 10%.
A major study published in Science on January 29, 2026, challenges that narrative fundamentally. Researchers led by Ben Shenhar at the Weizmann Institute of Science in Israel found that genetics account for approximately 54% of variation in human lifespan, more than double the previous consensus estimate. The finding doesn’t mean your habits are irrelevant. But it does mean the scientific community has been dramatically underestimating how much your DNA matters for how long you live, and the reasons for that underestimation reveal something important about how we study aging itself.
What the Study Found
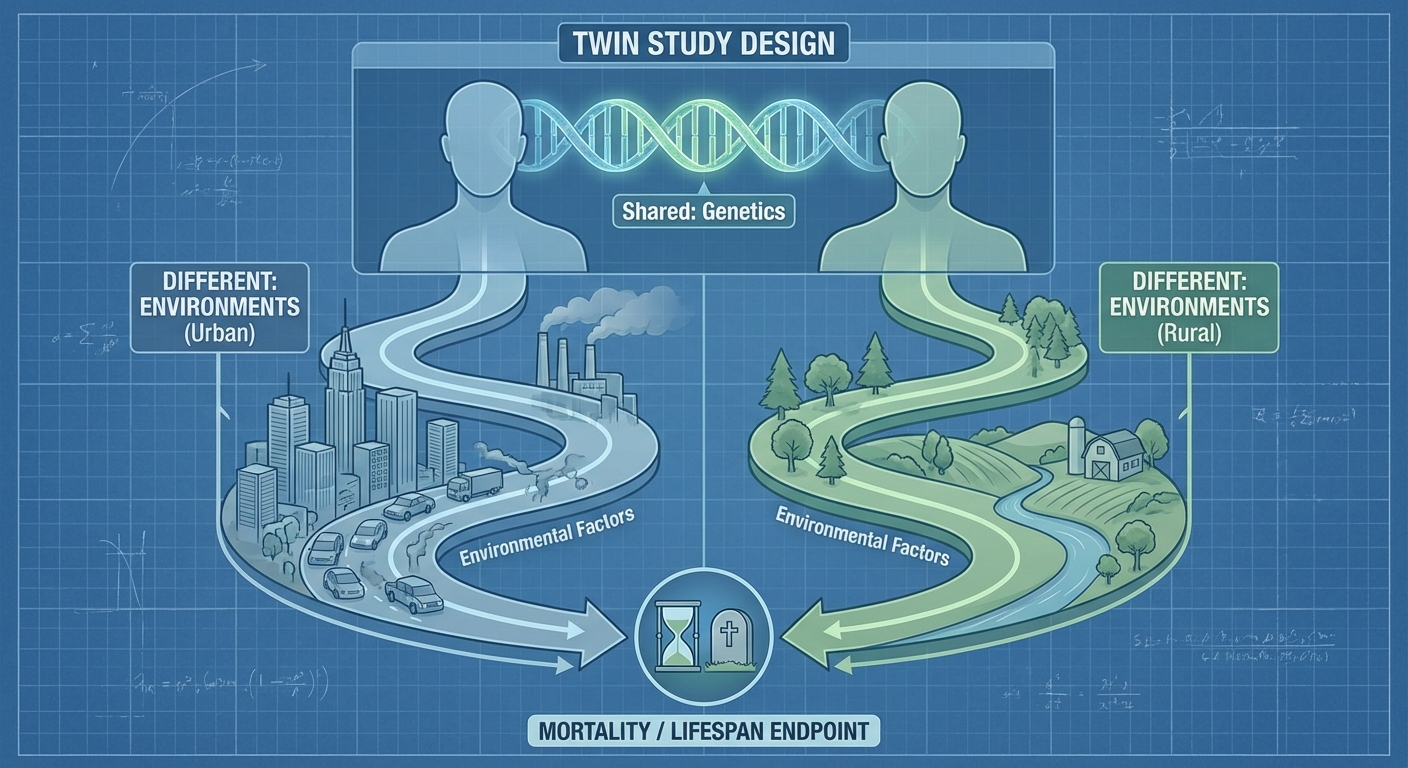
The research team, including collaborators at the Karolinska Institutet in Sweden and Leiden University Medical Center in the Netherlands, analyzed data from three Scandinavian twin databases: the Danish Twin Registry (birth years 1870 to 1900), the Swedish Twin Study (birth years 1886 to 1923), and the Swedish Adoption/Twin Study of Aging, known as SATSA (birth years 1900 to 1935). The SATSA cohort was particularly valuable because it included 168 identical twins and 411 fraternal twins who were raised apart, allowing researchers to separate genetic influence from shared environment with unusual precision.
The team applied two independent mathematical mortality models, both achieving a fit of r-squared greater than 0.967, to estimate heritability after correcting for a confounding factor that previous studies had overlooked. Their central finding: the heritability of intrinsic human lifespan is approximately 54%, with a standard error of plus or minus 3%. Three independent calculation methods within the SATSA cohort converged on this figure to within a few percentage points, providing strong internal validation.
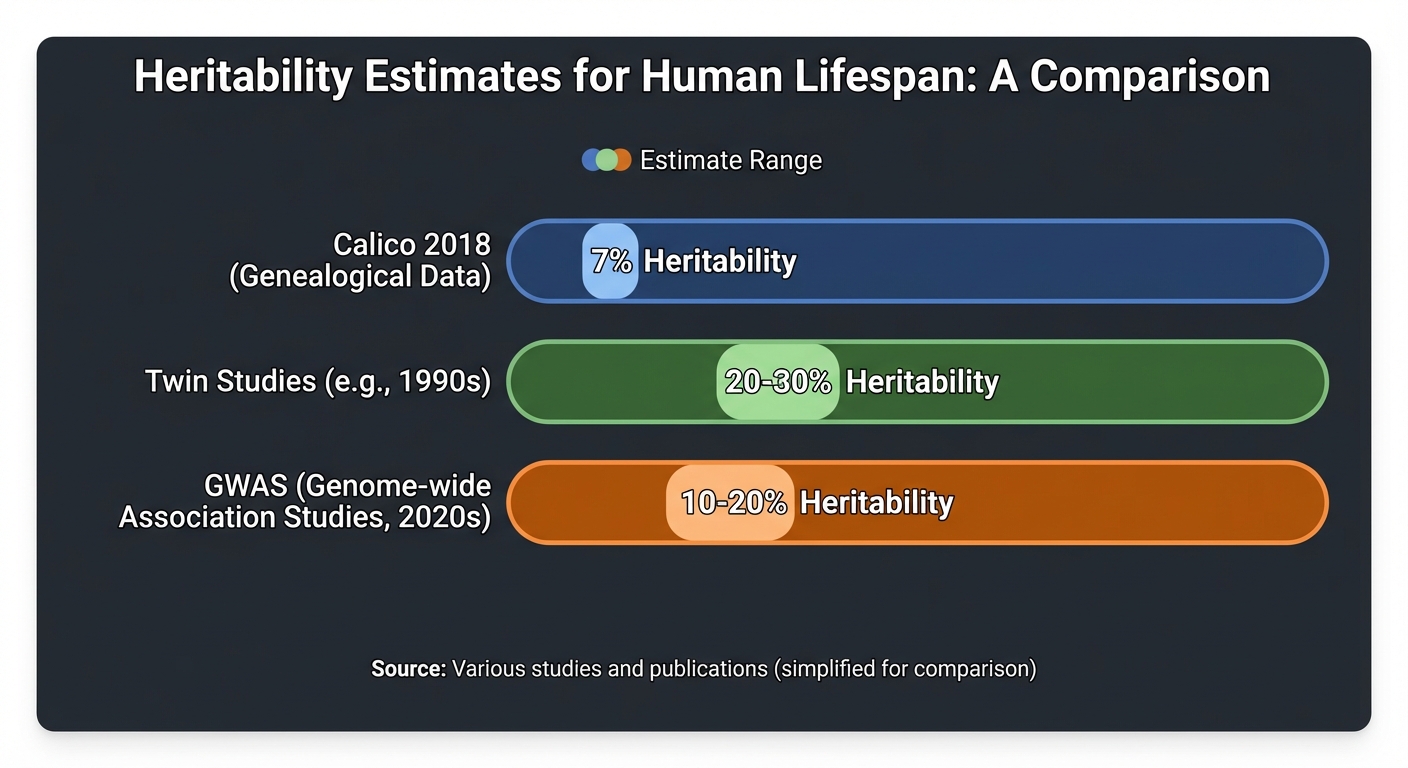
“For many years, human lifespan was thought to be shaped almost entirely by non-genetic factors, which led to considerable skepticism” about the role of genetics in aging, Shenhar noted. But the corrected estimate of roughly 50 to 55% actually aligns with what researchers find for most complex human traits. Height, body fat distribution, and muscle composition all show heritability around 50%. “The number that we got is not out of nowhere,” Shenhar said. “If you look at twin studies on pretty much anything in humans, you get this 50%.”
Uri Alon, the senior author and a professor at the Weizmann Institute’s Department of Molecular Cell Biology, emphasized the unique contribution of twins raised in separate environments. “Identical twins raised apart share their genes, but not their environment,” Alon explained. “This helps tease apart genetics from the environment.”
Why Previous Estimates Were So Low
The central methodological insight of the study is the distinction between extrinsic and intrinsic mortality. Extrinsic mortality refers to deaths caused by factors originating outside the body: accidents, homicides, infectious diseases, and environmental hazards. Intrinsic mortality refers to deaths from processes within the organism, including genetic mutations, age-related diseases, and gradual physiological decline. Previous heritability studies lumped both types of death together, and this created a systematic bias.
Before antibiotics and modern sanitation, extrinsic mortality was roughly ten times higher than it is today, primarily from infectious diseases like typhus, cholera, and tuberculosis. When a twin dies from cholera at age 30, that death tells you nothing about their genetic potential for longevity. But in a traditional heritability analysis, that death gets counted as evidence that genes don’t matter much, because the surviving twin who lived to 80 had the same DNA. The researchers showed mathematically that extrinsic mortality simultaneously reduces the measurable genetic variance between twin pairs while increasing the apparent environmental variance, making genes look far less important than they actually are.
“We neutralized external causes of death in existing databases,” Shenhar explained. By mathematically modeling and removing the effect of extrinsic mortality, the team revealed the genetic signal that had been buried under statistical noise for decades. As Alon put it, “We die much more of age-related diseases” today than in previous centuries, which means earlier studies based on historical data were systematically confounded by a world where infections and accidents killed people before their genetic aging programs could play out.
The finding received a dedicated commentary in the same issue of Science from Daniela Bakula and Morten Scheibye-Knudsen of the University of Copenhagen. They noted that the reframing “strengthens the rationale for large-scale efforts to identify longevity-associated variants, refine polygenic risk scores, and link genetic differences to specific biological pathways that regulate aging.” Scheibye-Knudsen pointed to the fact that species-level maximum lifespan is fundamentally genetic: humans live a maximum of roughly 120 years, yeast cells 13 days, and bowhead whales 200 years. The new estimate simply brings human within-species variation into line with this broader biological reality.
Genes and Lifestyle Are Not Opponents
The 54% heritability estimate raises an immediate question: if genes control more than half the game, does lifestyle still matter? The answer, supported by a companion study published in Science Advances, is an emphatic yes, but the relationship between genes and lifestyle is more nuanced than the popular “genes versus environment” framing suggests.
The Science Advances study, led by Yanling Lv at Huazhong University of Science and Technology, analyzed 103,649 UK Biobank participants over roughly a decade of follow-up. Researchers scored participants on both dietary quality (using five established healthy eating indices) and genetic longevity potential (using polygenic risk scores based on 19 genetic markers). The results showed that all five healthy dietary patterns reduced all-cause mortality risk by approximately 20%, while favorable genetics reduced mortality risk by about 15%.
Here is where the original analysis gets interesting. When you synthesize the two Science studies together, a picture emerges that is more subtle than either “genetics is destiny” or “lifestyle trumps genes.” The companion study found that the interaction between genetic inheritance and diet was “relatively weak,” meaning the benefits of healthy eating were essentially independent of your genetic hand. Whether you carry favorable or unfavorable longevity gene variants, a high-quality diet provided similar protection. In practical terms, a person with unfavorable longevity genetics who ate well gained nearly the same dietary benefit as a person with favorable genetics who ate well.
This independence is actually liberating. It means lifestyle interventions don’t need to overcome your genetic programming; they operate alongside it, through partially separate biological pathways. The life expectancy gains were substantial: men with both favorable genetics and the highest diet quality scores gained up to 3.2 additional years at age 45 compared to those with neither advantage, while women gained up to 5.5 years. Even moderate dietary adherence produced roughly half these benefits. The most impactful dietary component across all five indices was dietary fiber, with whole grains, fruits, and vegetables showing the strongest associations with longer life.
The takeaway isn’t that genes or lifestyle “wins.” Both contribute substantially, and crucially, you can influence the lifestyle half of the equation without needing to know what your genetic half looks like.
What This Means for the Longevity Movement
The revised heritability estimate has significant implications for how we approach aging research and longevity medicine. If genetics account for roughly half of lifespan variation, then the current scientific hunt for longevity-associated gene variants becomes far more justified, and potentially far more productive, than the field previously assumed.
Currently, only a handful of specific longevity genes have been identified: FOXO3, APOE, and SIRT6 among them. This is a strikingly small number given the new heritability estimate, a gap scientists call the “missing heritability” problem. The Shenhar study suggests that many more longevity variants exist but haven’t been found yet, likely because their individual effects are small and distributed across hundreds or thousands of genes. As the Science commentary noted, this creates a strong rationale for refining polygenic risk scores that could eventually allow precision medicine approaches to aging.
One particularly striking disease-specific finding from the study: genes influencing dementia account for approximately 70% of risk by age 80. This is considerably higher than the genetic contribution to cancer or heart disease, and it aligns with what researchers studying cognitive decline and brain health have observed about the stubborn biological roots of neurodegenerative disease. For anyone following the longevity field’s increasing attention to Alzheimer’s prevention, this statistic underscores both the challenge and the importance of genetic research in this space.
Not everyone in the field is fully convinced. Graham Ruby of Calico Life Sciences, whose 2018 study produced the much lower estimate, offered a nuanced counterpoint to STAT News. He argued that by controlling for extrinsic mortality, the Shenhar team “effectively changed the trait” being measured. In other words, “intrinsic lifespan” and “total lifespan” are different things, and the higher heritability applies specifically to the former. Eric Verdin of the Buck Institute for Research on Aging similarly cautioned that how you classify infection susceptibility (as extrinsic or partly genetic) affects the calculations, though he acknowledged that reanalysis still showed roughly 50% genetic contribution.
These are legitimate scientific debates about methodology, not challenges to the study’s core finding. The practical implication remains: the genetic contribution to aging is substantially larger than the research community assumed for the past 25 years, and this should redirect both research priorities and public understanding of what determines how long we live.
Beyond the 50/50 Split: Where Longevity Research Goes Next
The revised heritability picture suggests three areas where the longevity field may need to recalibrate. First, precision longevity medicine, already a growing field with companies offering wearable health monitoring and biological age testing, may eventually incorporate genetic longevity scores into their recommendations. If your polygenic risk score suggests below-average intrinsic longevity potential, the most evidence-based response isn’t fatalism; it’s doubling down on the lifestyle factors (diet, exercise, sleep, social connection) that operate through independent pathways.
Second, the pharmaceutical search for longevity drugs gains stronger justification. If specific gene variants extend lifespan, understanding the biological pathways they influence could reveal druggable targets. “If heritability is high, as we have shown, this creates an incentive to search for gene variants that extend lifespan, in order to understand the biology of aging and potentially to address it therapeutically,” Shenhar noted.
Third, public health messaging may need updating. The well-intentioned but oversimplified narrative that “lifestyle is 75 to 90% of longevity” has driven beneficial behavioral interventions, but it also sets unrealistic expectations. When someone with a family history of early death does everything “right” and still develops age-related disease in their 60s, the lifestyle-dominant model offers no explanation and considerable implicit blame. A more accurate model, one that acknowledges the roughly equal contributions of genes and environment, is both more honest and potentially more effective at motivating sustained healthy behaviors, because it frames lifestyle as genuinely impactful without promising genetic override.
The Evidence, Summarized
The Shenhar study represents one of the most important recalibrations in aging research in decades. By separating the noise of accidental and infectious deaths from the signal of biological aging, the team revealed that your DNA shapes roughly half of how long you’ll live, not the 10 to 25% that textbooks have cited for years. The finding is consistent with heritability estimates for virtually every other complex human trait, and it arrives at a moment when the longevity field is increasingly sophisticated enough to act on it.
Three things to take away. First, your genetic inheritance matters more than you’ve been told, but this doesn’t reduce your agency. The companion Science Advances study demonstrates that healthy eating benefits are largely independent of genetic longevity potential, meaning lifestyle interventions work regardless of your DNA. Second, if you have a family history of longevity or early death, take it more seriously as a data point about your own biological trajectory, not as destiny, but as context for how aggressively to pursue preventive health measures. Third, watch this space: the identification of longevity gene variants is likely to accelerate in the coming years, and polygenic risk scores for aging may eventually become as routine as cholesterol testing.
The study’s senior author put it simply: roughly half of longevity is genetic, and the other half is “everything else.” The good news is that “everything else” is entirely within your control.
Sources
- Heritability of intrinsic human life span is about 50% - Science, January 2026
- Rethinking the heritability of aging - Science Perspective, January 2026
- Longevity is in the genes: half of lifespan is heritable - Nature News, January 2026
- Genes influence human lifespan far more than thought - STAT News, January 2026
- Healthy dietary patterns, longevity genes, and life expectancy - Science Advances, 2026