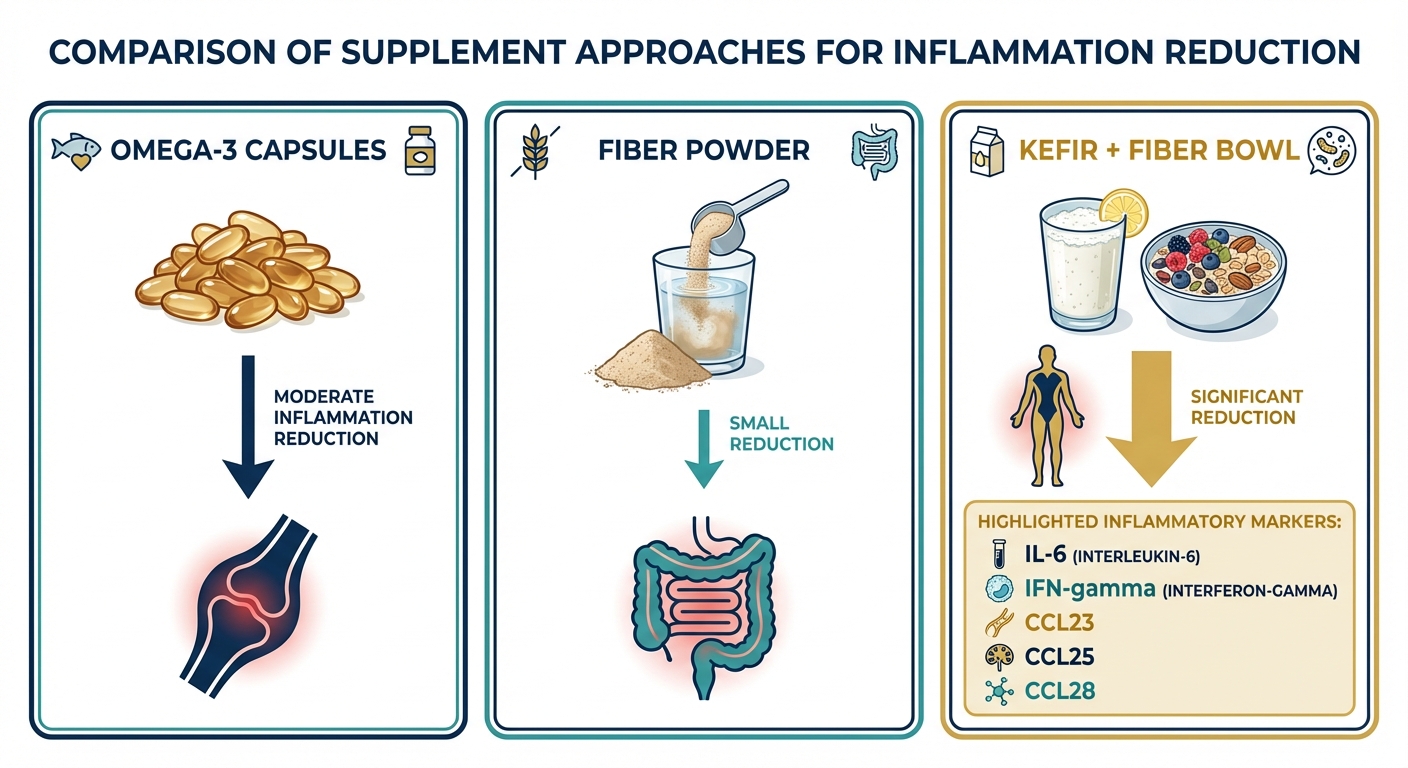
You take your omega-3 capsule every morning because you’ve heard it fights inflammation. But a new study from the University of Nottingham suggests you might be looking at the problem from the wrong end of your digestive tract. When researchers compared three common anti-inflammatory strategies head to head, a combination of fermented kefir and prebiotic fiber produced the broadest and most powerful reduction in inflammatory markers, outperforming both omega-3 supplements and fiber supplements taken alone.
The findings, published in the Journal of Translational Medicine in February 2026, add weight to a growing body of research suggesting that the most effective way to combat chronic inflammation isn’t popping a single supplement. It’s feeding the trillions of microbes already living in your gut and letting them do the heavy lifting. The implications could reshape how we think about inflammation management, shifting the focus from isolated nutrients to synergistic combinations that work with your body’s existing microbial ecosystem.
What the Study Found
The six-week trial, led by Dr. Amrita Vijay at the University of Nottingham’s School of Medicine, compared three dietary interventions in healthy adults: a synbiotic combination of naturally fermented goat’s milk kefir paired with a diverse prebiotic fiber blend, omega-3 fatty acid supplements, and inulin fiber supplements taken alone. Each group’s inflammatory status was measured using a comprehensive 96-protein inflammation panel, providing a far more detailed picture than the single-marker tests typical of older studies.
The results were striking. While all three interventions reduced some inflammatory markers, the synbiotic combination produced what the researchers described as “the most powerful and wide-ranging effects.” Adjusted analyses showed the kefir-fiber group achieved the largest number and greatest magnitude of significant protein decreases across the panel, including reductions in IL-6 (a key driver of chronic inflammation), IFN-gamma (an immune signaling molecule linked to autoimmune activity), SIRT2, 4EBP1, and several chemokines including CCL23, CCL25, and CCL28.
Beyond the inflammatory protein reductions, the synbiotic group showed metabolic improvements that neither comparison group achieved. Total cholesterol, LDL cholesterol, and non-HDL cholesterol all decreased in participants taking the kefir-fiber combination. These lipid changes suggest the anti-inflammatory benefits extended beyond immune modulation into cardiovascular risk reduction, a connection that aligns with what we know about inflammation’s role in atherosclerosis and heart disease.
“The synbiotic, combining fermented kefir with a diverse prebiotic fibre mix, had the most powerful and wide-ranging effects,” Dr. Vijay stated. “This suggests that the synergistic interaction between beneficial microbes and dietary fibres may be key to supporting immune balance.”
Why the Combination Works Better Than Either Alone
The synbiotic advantage comes down to a principle that’s deceptively simple: probiotics need fuel, and prebiotics need someone to eat them. When you take an omega-3 supplement, you’re delivering an anti-inflammatory compound directly into your bloodstream. It works, but it operates through a single pathway, primarily by competing with pro-inflammatory omega-6 fatty acids for incorporation into cell membranes and by serving as precursors to specialized pro-resolving mediators.
When you take fiber alone, you’re providing food for your gut bacteria, but you’re relying on whatever microbial population happens to be present. If your microbiome is already depleted or imbalanced (a common condition in Western diets), fiber alone may not produce optimal results because the bacteria best equipped to ferment it aren’t there in sufficient numbers.
The synbiotic approach solves both problems simultaneously. Fermented kefir delivers a diverse community of live probiotic bacteria and yeasts directly into the gut, while the prebiotic fiber blend (which in this study contained 18 different natural food-source fibers) provides the specific substrates those microbes need to thrive and multiply. The result is a flourishing microbial ecosystem that produces beneficial metabolites at volumes neither intervention could achieve independently.
The key metabolite driving much of the anti-inflammatory effect appears to be butyrate, a short-chain fatty acid produced when gut bacteria ferment fiber. The study found that serum butyrate concentrations increased significantly in the synbiotic group, and these increases correlated inversely with IL-6 levels, meaning higher butyrate meant lower inflammation. Butyrate is recognized for its anti-inflammatory and immune-regulating effects throughout the body, from strengthening the gut barrier to modulating immune cell behavior in distant tissues.
The Chronic Inflammation Problem
Understanding why this research matters requires grasping the scope of chronic low-grade inflammation in modern health. Unlike the acute inflammation that causes a sprained ankle to swell (a healthy, protective response), chronic systemic inflammation operates silently for years or decades, gradually damaging tissues and driving the progression of nearly every major chronic disease.
Heart disease, type 2 diabetes, Alzheimer’s disease, certain cancers, autoimmune conditions, and even depression have all been linked to elevated inflammatory markers. The World Health Organization has identified chronic inflammatory diseases as the most significant cause of death worldwide, with more than 50% of all deaths attributable to inflammation-related conditions including stroke, chronic respiratory diseases, heart disorders, and cancer.
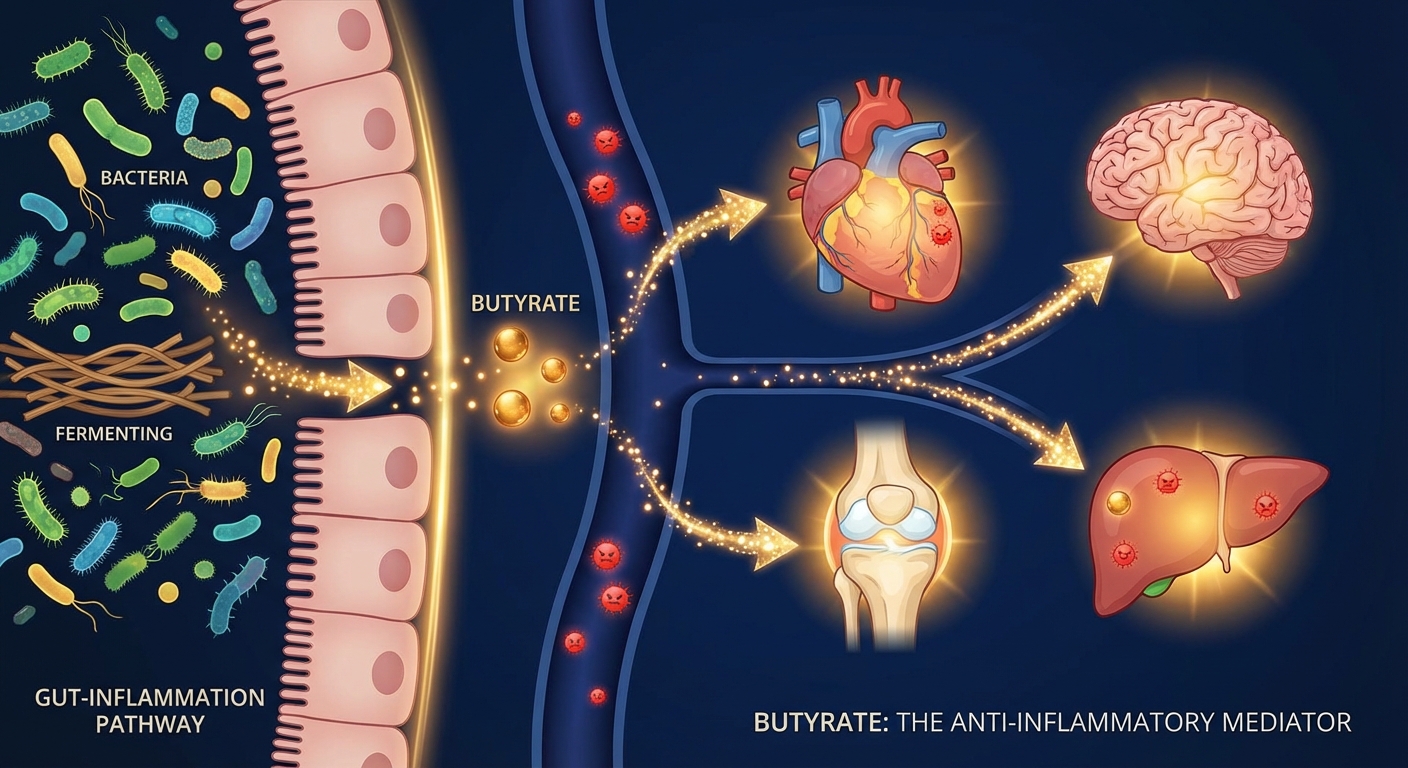
What makes the Nottingham study particularly relevant is its focus on this systemic dimension. The 96-protein panel didn’t just measure one or two markers in isolation. It captured a comprehensive snapshot of the inflammatory landscape across multiple pathways. The fact that the synbiotic reduced markers across such a broad range suggests it’s modulating inflammation at a fundamental level rather than simply suppressing a single pathway, which is exactly what you’d expect from an intervention that works through the microbiome, a system that interfaces with virtually every aspect of immune function.
For readers who have been following the fibermaxxing trend that’s gained momentum in 2026, this study offers a critical upgrade to the conversation. Fiber alone is good. Fiber paired with the right microbial partners is substantially better.
An Original Framework: The Synbiotic Hierarchy
Most supplement recommendations treat anti-inflammatory interventions as interchangeable: take omega-3, or eat more fiber, or try probiotics. This study suggests we need a more nuanced framework, what I’d call the Synbiotic Hierarchy of anti-inflammatory effectiveness.
At the base level, you have single-pathway interventions like omega-3 supplements. These deliver a specific anti-inflammatory compound through a defined biochemical mechanism. They work, and decades of research support their benefits, but they’re limited to the pathways they can directly influence. Our recent coverage of fish oil research and the ALOX15 gene highlighted how individual genetic variation can further limit omega-3 effectiveness, meaning the same dose that benefits one person may do little for another.
The middle tier includes single-component microbiome interventions: probiotics alone or prebiotics alone. These engage the gut ecosystem but only partially. Probiotics without appropriate fiber may not colonize effectively. Fiber without diverse bacteria may ferment incompletely or produce suboptimal metabolite profiles.
At the top sits the synbiotic approach, which simultaneously introduces beneficial organisms and their preferred fuel sources. This creates what microbiologists call a “competitive advantage” for anti-inflammatory bacterial strains, allowing them to outcompete potentially harmful species while maximizing metabolite production. The Nottingham data suggests this hierarchical model holds: synbiotic outperformed omega-3, which outperformed fiber alone for most inflammatory markers.
This framework has practical implications for anyone managing chronic inflammation. Rather than debating whether to take fish oil or a probiotic, the evidence increasingly points toward layered strategies that address the microbiome as an ecosystem rather than targeting individual biochemical pathways.
How to Build Your Own Synbiotic Protocol
The study used naturally fermented goat’s milk kefir combined with 18 different prebiotic fiber sources. While the exact commercial product formulation isn’t publicly available, you can approximate the approach with whole foods and strategic supplementation.
Fermented foods that deliver diverse probiotic strains:
- Kefir (dairy or water-based): Contains 30-50 different bacterial and yeast strains, far more diverse than most commercial probiotics
- Sauerkraut (unpasteurized): Rich in Lactobacillus species
- Kimchi: Provides Lactobacillus, Leuconostoc, and Weissella species
- Miso and tempeh: Offer different microbial profiles from Asian fermentation traditions
Prebiotic fiber sources to diversify your microbial fuel supply:
- Jerusalem artichokes: Among the richest sources of inulin (approximately 18g per 100g)
- Chicory root: Another concentrated inulin source, commonly used in fiber supplements
- Garlic and onions: Contain fructooligosaccharides (FOS) that selectively feed beneficial Bifidobacteria
- Green bananas and plantains: High in resistant starch, a prebiotic that produces significant butyrate
- Oats: Provide beta-glucan, a prebiotic fiber with its own anti-inflammatory properties
- Legumes: Deliver diverse fiber types including resistant starch and galactooligosaccharides

The critical insight from the study is diversity. The prebiotic component used 18 different fiber sources, not just a single type like inulin. This matters because different bacterial species prefer different fiber substrates. A diverse fiber intake supports a diverse microbiome, and microbiome diversity is consistently associated with better health outcomes across dozens of studies. Aim for at least 30 different plant foods per week, a target endorsed by the American Gut Project’s research, to maximize the range of fibers reaching your gut bacteria.
What This Doesn’t Mean
Before you throw out your fish oil capsules, some important caveats deserve attention. The study was conducted in healthy adults over six weeks. We don’t yet know whether the same results would hold in people with existing inflammatory conditions like rheumatoid arthritis, inflammatory bowel disease, or cardiovascular disease. Dr. Vijay’s team has indicated that testing on individuals with specific health conditions is the next research phase.
The study also did not directly measure microbiome composition, meaning the researchers observed the downstream effects (reduced inflammatory proteins, increased butyrate) without confirming exactly which bacterial species were responsible. This is an important limitation because it means the mechanistic pathway, while biologically plausible, remains partially inferential.
Additionally, omega-3 fatty acids have benefits that extend well beyond inflammation reduction, including structural roles in brain cell membranes, cardiovascular protection through triglyceride reduction, and potential benefits for mood disorders. The Nottingham study measured one specific dimension (inflammatory protein reduction) where the synbiotic excelled, but this doesn’t invalidate the broader evidence supporting omega-3 supplementation for other health outcomes.
Finally, the goat’s milk kefir used in the study is a specific product with a particular microbial profile. Commercial kefir products vary enormously in their bacterial diversity and viability. Store-bought kefir that has been pasteurized after fermentation may contain few or no live organisms. For maximum benefit, look for products labeled “live and active cultures” or, better yet, make your own using kefir grains, which are widely available online and produce a more diverse microbial community than commercial starters.
Your Gut Health Action Plan
This study reinforces a principle that’s becoming increasingly clear in nutritional science: the most powerful dietary interventions work with your body’s microbial ecosystem, not around it. Rather than relying solely on isolated supplements to fight inflammation, building a synbiotic approach that pairs fermented foods with diverse fiber sources appears to produce broader and more robust anti-inflammatory effects.
Start with these specific steps. First, introduce a daily serving of fermented kefir (approximately 200-250ml), preferably made from live kefir grains. Second, diversify your fiber intake beyond the single-source supplements by incorporating at least five different prebiotic foods into your weekly rotation. Third, maintain your existing omega-3 intake if you’re taking it for cardiovascular or cognitive benefits, since the synbiotic approach complements rather than replaces other evidence-based interventions.
The research team at Nottingham plans to expand their work to clinical populations with existing inflammatory conditions. Until those results arrive, the current evidence offers a compelling case for rethinking the single-supplement approach to inflammation and embracing the synbiotic strategy that your gut bacteria have been waiting for.
Sources
- This kefir and fiber combo beat omega-3 in slashing inflammation - ScienceDaily, February 2026
- Kefir plus prebiotic blend beats other supplements for reducing inflammation - University of Nottingham News
- Combination of Kefir and Prebiotic Fiber Could Lower Chronic Inflammation Risk - Technology Networks
- Fermented Kefir-Fiber Synbiotic Shows Broad Anti-Inflammatory Protein Reductions in Healthy Adults - ReachMD






