Your heart beats roughly 100,000 times per day. Each contraction requires precise coordination between millions of muscle cells, each powered by tiny energy factories called mitochondria. For the 1.2 million Australians living with type 2 diabetes, and the estimated 38 million Americans in the same situation, those cellular power plants are under siege. New research from the University of Sydney reveals that diabetes doesn’t just increase the statistical risk of heart disease. It physically rewires the heart’s structure and fundamentally alters how cardiac muscle produces and uses energy.
The findings, published in EMBO Molecular Medicine in January 2026, help explain a medical mystery that has puzzled cardiologists for decades: why do people with diabetes develop heart failure at rates far exceeding what traditional risk factors would predict? The answer, it turns out, lies in molecular changes that begin long before symptoms appear.
The Hidden Molecular Damage
Dr. Benjamin Hunter and Associate Professor Sean Lal from the University of Sydney’s School of Medical Sciences examined heart tissue from patients with diabetic ischemic cardiomyopathy, the leading cause of heart failure. What they found was striking. Diabetes doesn’t simply stress the heart through high blood sugar levels. It triggers a cascade of molecular changes that compromise the organ’s fundamental ability to function.
The research team identified three primary mechanisms of damage. First, diabetes reduces insulin sensitivity in glucose transporters, the cellular doorways that allow heart cells to take in glucose for fuel. When these transporters malfunction, cardiac cells can’t efficiently access their primary energy source. Second, this glucose deprivation places enormous stress on mitochondria, forcing them to work harder with less fuel. Third, the structural proteins responsible for heart muscle contraction begin to degrade, while stiff fibrous tissue accumulates in their place.
“Diabetes alters how the heart produces energy, maintains its structure under stress, and contracts to pump blood,” Dr. Hunter explained. This isn’t gradual wear and tear. It’s active molecular remodeling that weakens the heart from the inside out.

Why Energy Production Matters More Than You Think
Your heart is the most metabolically active organ in your body. At rest, it consumes more oxygen per gram of tissue than any other organ, and it can’t take breaks. Unlike skeletal muscle, which can switch to anaerobic metabolism during brief intense efforts, cardiac muscle requires a constant supply of ATP, the cellular energy currency, to keep beating.
Under normal circumstances, the heart is remarkably flexible in its fuel choices. It can burn fatty acids, glucose, lactate, and ketones depending on what’s available. This metabolic flexibility is crucial for adapting to different conditions, whether you’re sleeping, sprinting, or sitting through a long meeting. Diabetes compromises this flexibility by impairing glucose uptake, forcing the heart to rely more heavily on fatty acid oxidation.
The problem is that burning fatty acids requires more oxygen than burning glucose. In a heart already stressed by high blood pressure, arterial stiffness, or coronary artery disease, common companions to diabetes, this increased oxygen demand can tip the balance toward energy deficit. The mitochondria struggle to keep up. Reactive oxygen species accumulate. Cellular damage accelerates.
This explains something clinicians have observed for years: people with diabetes often develop heart failure even when their coronary arteries look relatively clear on imaging. The damage isn’t primarily in the blood vessels. It’s in the heart muscle cells themselves.
The Structural Collapse
Beyond energy production, the Sydney research revealed significant changes to the proteins that give heart muscle its contractile power. Calcium handling, essential for the precisely timed contractions that pump blood, becomes disrupted. The proteins that form the structural scaffolding of cardiac cells show reduced expression.
Perhaps most concerning is the accumulation of fibrous tissue within the heart muscle. In healthy hearts, small amounts of connective tissue provide structural support. In diabetic hearts, this fibrosis becomes excessive, creating stiff patches that can’t contract properly. The result is a heart that’s simultaneously weaker and stiffer, struggling both to pump blood out and to relax enough to fill properly.
This combination, known as diabetic cardiomyopathy, represents a distinct disease process separate from the coronary artery disease that diabetes also promotes. You can have pristine arteries and still develop heart failure from diabetes alone. The Sydney findings suggest this progression may be more common than previously recognized.

What This Means for Prevention and Treatment
The implications of this research extend beyond understanding the problem. If diabetes physically remodels the heart at the molecular level, then prevention strategies need to target these specific mechanisms, not just blood sugar numbers.
Associate Professor Lal noted that the findings “could be used to inform diagnosis criteria and disease management strategies across cardiology and endocrinology.” Currently, diabetic cardiomyopathy often goes undiagnosed until symptoms become severe. Standard echocardiograms may miss early changes because they primarily assess pumping function, not the underlying molecular health of cardiac tissue.
Advanced imaging techniques like cardiac MRI with strain analysis or T1 mapping can detect fibrosis earlier. Biomarkers indicating mitochondrial stress or collagen turnover might eventually allow screening through simple blood tests. The goal would be identifying diabetic heart damage in its earliest, most treatable stages.
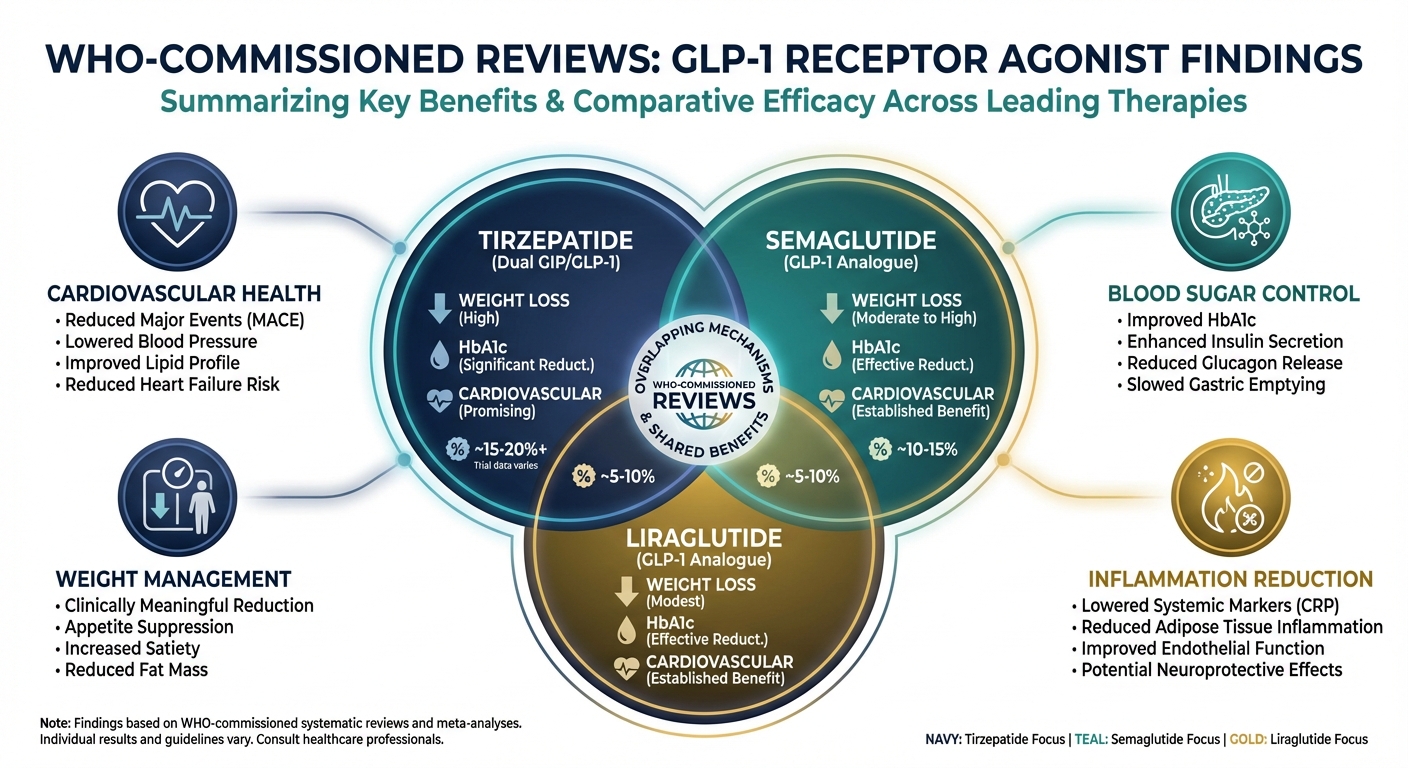
Treatment approaches may also need refinement. The newer GLP-1 receptor agonists and SGLT2 inhibitors, originally developed for blood sugar control, have shown remarkable heart protection benefits that go beyond glucose management. Research suggests these medications may directly improve mitochondrial function and reduce cardiac fibrosis, addressing the mechanisms identified in the Sydney study.
For those managing type 2 diabetes, this research underscores why cardiovascular protection deserves as much attention as blood sugar targets. Your A1C numbers matter, but so does the metabolic health of every cell in your heart.
The Exercise Connection
One bright spot in this otherwise sobering research is the potential for exercise to counteract some of these changes. Regular aerobic exercise improves mitochondrial function, enhances metabolic flexibility, and may even reduce cardiac fibrosis. Studies have shown that structured exercise programs can improve heart function in people with diabetic cardiomyopathy, though the optimal prescription remains under investigation.
The key appears to be consistency over intensity. Moderate aerobic exercise performed regularly, think brisk walking, cycling, or swimming for 150 minutes weekly, stimulates mitochondrial biogenesis and improves insulin sensitivity in both skeletal muscle and cardiac tissue. High-intensity interval training shows promise for those who can tolerate it, but the foundation is building and maintaining an active lifestyle.
Resistance training also plays a role by improving overall metabolic health and glucose disposal, reducing the burden on the heart’s compromised glucose uptake systems. The combination of aerobic and resistance exercise, performed consistently over months and years, may help preserve cardiac function even as diabetes management presents ongoing challenges. Understanding your heart rate variability can help guide appropriate exercise intensity.

Beyond Blood Sugar: A Whole-System Approach
The Sydney research fits into a broader emerging understanding of diabetes as a systemic metabolic disease affecting virtually every organ. The gut-inflammation connection in diabetes shows how intestinal health influences diabetic complications. Nighttime light exposure affects both heart health and glucose regulation through circadian disruption. Each piece of the puzzle reinforces a central theme: managing diabetes effectively requires addressing multiple interconnected systems.
Blood sugar control remains important, but it’s no longer sufficient as a sole focus. Protecting the heart means optimizing sleep, managing stress, maintaining physical activity, controlling blood pressure, and addressing inflammation. The molecular damage identified in the Sydney study doesn’t happen in isolation. It develops in the context of a metabolic environment shaped by dozens of daily choices and health factors.
For people living with type 2 diabetes, this complexity can feel overwhelming. The practical takeaway is actually simpler than it might seem: the same interventions that improve metabolic health generally also protect the heart. Regular movement, adequate sleep, stress management, whole food nutrition, and appropriate medication when indicated all work synergistically. You don’t need to address each mechanism individually. You need to create an environment where your body can heal.
The Bottom Line
Type 2 diabetes fundamentally changes how your heart produces energy, maintains its structure, and pumps blood. New research from the University of Sydney shows that diabetes triggers molecular remodeling that can lead to heart failure even without significant coronary artery disease. These changes, including impaired glucose uptake, mitochondrial stress, protein degradation, and fibrous tissue accumulation, begin before symptoms appear and progress silently.
The findings emphasize that heart protection should be central to diabetes management, not an afterthought. Early detection through advanced imaging, medications that target cardiac mechanisms directly, and lifestyle interventions focused on mitochondrial health all play roles in prevention.
Next Steps:
- Discuss cardiac screening with your healthcare provider if you have type 2 diabetes, even without symptoms
- Prioritize consistent moderate exercise (150+ minutes weekly) to support mitochondrial function
- Ask about SGLT2 inhibitors or GLP-1 agonists if not already part of your treatment plan
- Monitor blood pressure and manage stress, both affect cardiac remodeling
- Focus on metabolic health holistically, not just blood sugar numbers
Sources: EMBO Molecular Medicine (January 2026), University of Sydney School of Medical Sciences, Dr. Benjamin Hunter and Associate Professor Sean Lal research, American Heart Association cardiovascular-diabetes guidelines.