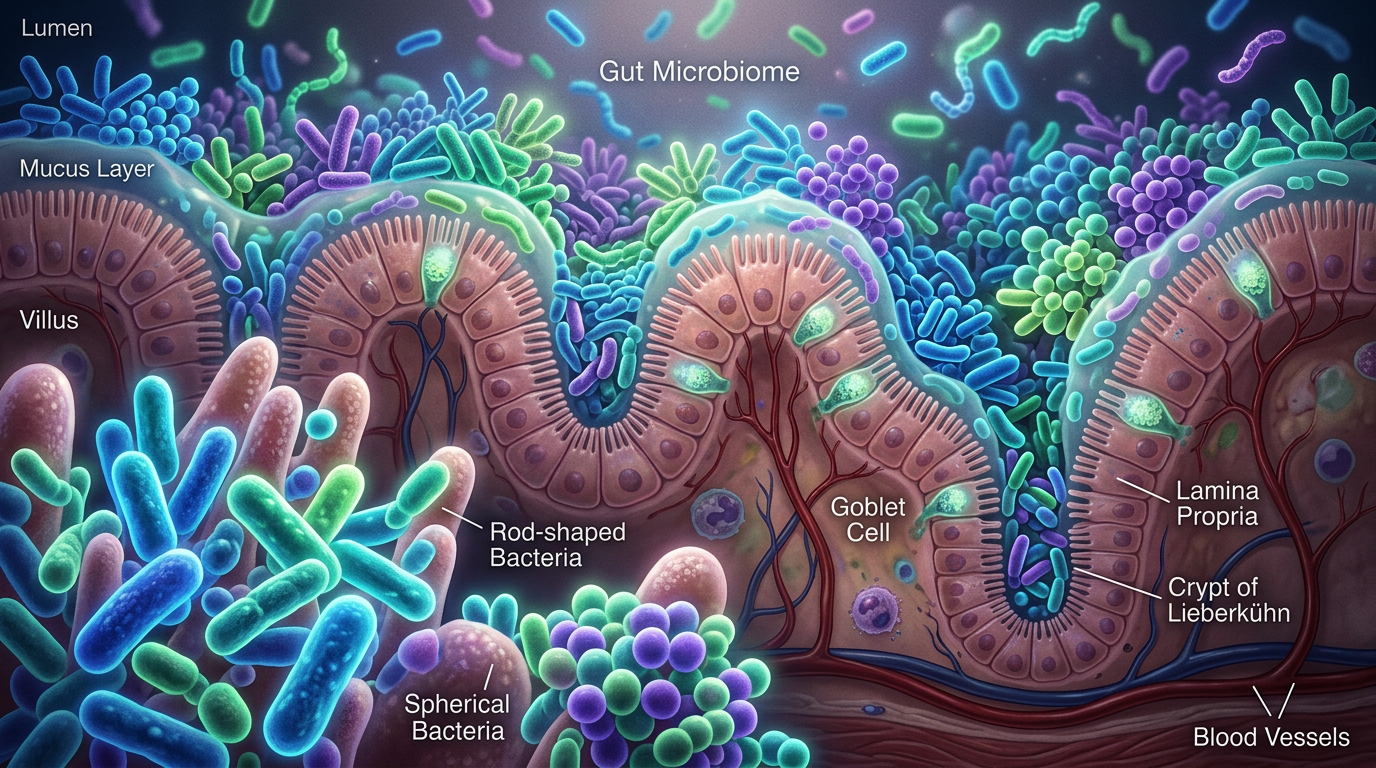
Somewhere in your gut right now, trillions of bacteria are producing vitamins, breaking down fibers, and communicating with your immune system. Scientists have spent decades cataloging the most well-known players in this ecosystem, from Lactobacillus to Bifidobacterium, and building an entire probiotic industry around them. But a new study published in Cell Host & Microbe reveals that some of the most important bacteria in the human gut have been hiding in plain sight. Researchers at the University of Cambridge analyzed gut microbiome samples from more than 11,000 people across 39 countries, and they found that a little-studied group of microbes called CAG-170 shows up consistently in healthy individuals while appearing at significantly lower levels in people living with chronic diseases. The finding suggests that the probiotic supplements lining pharmacy shelves may be targeting the wrong bacteria entirely.
A Global Search Across 39 Countries
The research team, led by Dr. Alexandre Almeida of the University of Cambridge’s Department of Veterinary Medicine, did not collect new stool samples or recruit volunteers. Instead, they turned to an enormous existing database: the Unified Human Gastrointestinal Genome catalogue. This resource contains genetic information from gut microbiome samples collected by researchers around the world over many years, spanning populations across Europe, North America, and Asia. By mining this dataset with advanced computational techniques, Almeida’s team could look for patterns that no single regional study would have the statistical power to detect.
The analysis unfolded in three stages. First, the team compared microbiome data from 11,115 individuals across 39 countries, looking for bacterial groups that appeared more frequently in healthy people than in those with diagnosed conditions. Second, they ran a separate computational analysis on samples from more than 6,000 healthy individuals to confirm their findings. Third, they specifically measured CAG-170 levels in people experiencing dysbiosis, a state where the gut microbiome has fallen out of balance. All three lines of evidence pointed in the same direction: CAG-170 bacteria were consistently more abundant in people who were healthy.
“These are a fundamental and underappreciated component of human health,” Almeida said of the CAG-170 group. The bacteria turned up at lower levels in people with 13 different chronic conditions, including inflammatory bowel disease, Crohn’s disease, obesity, chronic fatigue syndrome, colorectal cancer, Parkinson’s disease, multiple sclerosis, irritable bowel syndrome, rheumatoid arthritis, anxiety, and depression. That range is striking because these conditions involve very different organ systems, from the gut to the brain to the joints, suggesting that CAG-170 may play a foundational role in overall health rather than being tied to any single disease pathway.
What CAG-170 Actually Does in Your Gut
The Cambridge researchers did not just observe where CAG-170 bacteria live. They also investigated what these microbes are doing. By examining the genetic material of CAG-170, the team identified two primary functions. First, these bacteria produce large quantities of vitamin B12, a nutrient that humans need for nerve function, red blood cell production, and DNA synthesis. Second, CAG-170 carries a suite of enzymes that break down complex carbohydrates, sugars, and dietary fibers, essentially helping the body extract nutrients from plant-based foods that would otherwise pass through undigested.
The vitamin B12 finding comes with an important nuance. While humans absorb most of their B12 in the upper small intestine, CAG-170 bacteria reside further along the digestive tract, in the large intestine. That means the B12 they produce likely does not get absorbed by the human host directly. Instead, it probably feeds other beneficial bacteria in the gut ecosystem, acting as a kind of microbial currency that sustains a healthy community of microorganisms. Research published in Frontiers in Nutrition has confirmed that healthy adult gut microbiota can sustain their own B12 requirements through in-house microbial production, and CAG-170 appears to be one of the primary contributors to this internal supply chain.
“CAG-170 bacteria appear to be key players in human health, likely by helping us to digest food and keeping the whole microbiome running smoothly,” Almeida explained. This framing matters because it shifts the conversation away from bacteria that directly benefit humans and toward bacteria that keep the entire microbial ecosystem functioning. If CAG-170 declines, the ripple effects could starve other beneficial species of the B12 and breakdown products they depend on, potentially triggering a cascade of microbial imbalances.
The Probiotic Industry’s Blind Spot
The global probiotic market is worth more than $60 billion, yet most commercial probiotic supplements still rely on bacterial strains identified decades ago. Lactobacillus and Bifidobacterium dominate supplement shelves not because they are necessarily the most important bacteria in the human gut, but because they were among the earliest species scientists managed to grow in laboratory cultures. CAG-170 has remained obscure for a simple reason: most of its species cannot currently be grown in a lab. The Cambridge team identified these bacteria entirely through metagenomics, a technique that reads all the microbial DNA in a sample at once and then uses software to separate out individual species.
This distinction between what we can culture and what we can detect genetically is one of the most significant gaps in gut health science. Traditional microbiology required growing bacteria on petri dishes to study them, which meant that any species that refused to grow under lab conditions went essentially unnoticed. Modern sequencing technology has revealed that the vast majority of gut bacteria fall into this “unculturable” category. CAG-170 is a prime example: it has been sitting in human guts around the world this entire time, contributing to health in meaningful ways, yet it escaped scientific attention until computational methods caught up.
The implications for the probiotic industry are significant. If CAG-170 cannot be cultured, it cannot be put into a capsule, at least not yet. But the findings suggest that the next generation of probiotic supplements will need to look beyond the familiar species. Several research groups are already working on techniques to culture previously unculturable bacteria, and the identification of CAG-170 as a potential health marker gives those efforts a concrete target. In the meantime, the most practical approach may be to support CAG-170 indirectly, by feeding it the complex carbohydrates and fibers it depends on.

From Correlation to Causation: What We Still Don’t Know
The Cambridge study is observational, which means it establishes an association between CAG-170 levels and health status without proving that one causes the other. It is entirely possible that chronic diseases reduce CAG-170 levels rather than the other way around. Inflammation from conditions like Crohn’s disease or ulcerative colitis, for example, can alter the gut environment in ways that make it hostile to certain bacterial species. In that scenario, low CAG-170 would be a consequence of disease rather than a contributing cause.
Dr. Almeida and his colleagues acknowledged this limitation directly. The study’s three-part design, analyzing healthy populations separately from diseased populations and then checking for dysbiosis specifically, was intended to strengthen the correlation, but it cannot replace the kind of controlled intervention studies that would be needed to establish causation. Those studies would require either adding CAG-170 to the guts of people with chronic disease (difficult, given the culturing problem) or selectively removing it from healthy people (ethically problematic and technically complex).
There is also the question of confounding variables. People who are healthy tend to eat more diverse diets, exercise more frequently, sleep better, and experience less chronic stress. All of these factors independently affect the gut microbiome. The consistent appearance of CAG-170 across 39 countries and diverse populations makes the correlation more robust than a single-country study could, but it does not eliminate the possibility that CAG-170 is a marker of healthy behavior rather than a driver of health itself. Future research will need to untangle these threads, and that work is already underway at several institutions studying the gut microbiome.
Original Analysis: Why This Discovery Marks a Turning Point for Microbiome Science
The identification of CAG-170 fits into a larger pattern that has been building in microbiome research for the past decade. When the Human Microbiome Project launched in 2007, scientists expected to find a relatively small set of “good” and “bad” bacteria that would explain most health outcomes. Instead, they found staggering complexity: thousands of species interacting in ways that defied simple categorization. The early probiotic industry latched onto the few species that were easy to culture and market, creating a market where the same handful of Lactobacillus strains appear in product after product regardless of the condition they claim to address.
What makes the CAG-170 discovery different from previous microbiome findings is its combination of global consistency and functional specificity. Many earlier studies identified bacterial groups associated with health in specific populations, say, fiber-fermenting bacteria in rural African communities or B12 producers in Japanese populations with seaweed-heavy diets. CAG-170, by contrast, showed up across 39 countries spanning three continents, suggesting it is not a product of any particular dietary pattern but rather a fundamental component of a well-functioning human gut. This kind of cross-cultural consistency is rare in microbiome research and lends the finding a weight that single-population studies cannot match.
The discovery also raises a provocative question about how we measure gut health. Currently, most clinical microbiome tests focus on overall diversity, the idea that more species equals better health. But the CAG-170 finding suggests that specific bacterial groups may matter more than total diversity. A person could have high microbial diversity but still lack adequate CAG-170, and the Cambridge data suggests they would be at higher risk for chronic disease. This challenges the “diversity is everything” framework and pushes toward a more nuanced model where certain keystone species, bacteria that support the broader ecosystem, deserve special attention. The parallel to ecology is instructive: in forest ecosystems, the loss of a single keystone species like wolves can trigger cascading collapse even when overall species counts remain high. CAG-170 may function as a keystone species in the gut ecosystem, and its decline may set off similar cascading effects that eventually manifest as chronic disease.
This reframing also connects to the growing “fibermaxxing” trend in nutrition. If CAG-170 thrives on complex carbohydrates and dietary fibers, then the recent surge of interest in high-fiber diets takes on added scientific weight. The trend has largely been driven by general research linking fiber intake to lower disease risk, but CAG-170 provides a specific mechanistic explanation: fiber feeds the bacteria that produce B12 for the rest of the microbial community, which in turn supports immune regulation, nutrient absorption, and metabolic health. It is one of the clearest examples yet of how dietary choices translate into specific microbial outcomes.
How to Support Your Gut Bacteria Today
While researchers work to culture CAG-170 and develop targeted interventions, the existing evidence points toward several practical steps that may help maintain healthy levels of these bacteria. The logic is straightforward: since CAG-170 breaks down complex carbohydrates and fibers, eating more of those foods should support its growth. Since CAG-170 produces B12 for the broader microbial community, supporting overall gut ecosystem diversity should create conditions where CAG-170 can thrive.
Here are evidence-based strategies drawn from the broader microbiome literature:
- Prioritize diverse plant fibers. Eat at least 30 different plant foods per week. Each type of fiber feeds different bacterial species, and diversity of inputs leads to diversity of outputs. Root vegetables, legumes, whole grains, and cruciferous vegetables are all sources of the complex carbohydrates that CAG-170’s enzymes target.
- Include fermented foods regularly. Kefir, yogurt, sauerkraut, kimchi, and miso introduce live bacteria and create conditions that support existing beneficial species. A Stanford study found that fermented foods combined with prebiotic fibers produced greater reductions in inflammatory markers than either approach alone.
- Minimize unnecessary antibiotics. Broad-spectrum antibiotics can wipe out entire bacterial populations indiscriminately, and unculturable species like CAG-170 may be particularly slow to recover because they cannot be reintroduced through supplements.
- Limit ultra-processed foods. Diets high in emulsifiers, artificial sweeteners, and refined carbohydrates have been shown to reduce microbial diversity and shift the gut environment toward inflammatory species.
These steps are not specific to CAG-170, and that is actually the point. Until scientists can develop targeted interventions for this particular bacterial group, the best approach is to create gut conditions that support the widest possible range of beneficial microbes. The evidence linking diet quality to long-term health outcomes continues to strengthen, and findings like the Cambridge study provide a biological mechanism that helps explain why.

What the Research Tells Us
The discovery of CAG-170 is a reminder that the human gut microbiome still contains surprises. Despite more than two decades of intensive study, entire groups of bacteria with significant health implications have gone unnoticed because they resisted the traditional tools of microbiology. The Cambridge team’s computational approach revealed not just a new bacterial group but a potential new framework for understanding why some people develop chronic diseases and others do not. Across 39 countries and 13 disease conditions, the pattern held: people with more CAG-170 were healthier.
That does not mean you should panic about your CAG-170 levels. There is currently no clinical test for these bacteria, no supplement that contains them, and no way to know where you fall on the spectrum. What the research does suggest is that the fundamentals of gut health, eating diverse fibers, consuming fermented foods, avoiding unnecessary disruptions to your microbiome, may be even more important than previously understood. These habits do not just feed the bacteria we already know about. They support the ones we are only beginning to discover.
The next chapter of this research will likely focus on two fronts: developing methods to culture CAG-170 in the laboratory, which would open the door to targeted probiotic development, and conducting longitudinal studies to determine whether changes in CAG-170 levels precede or follow the onset of chronic disease. Both efforts will take years. In the meantime, the practical takeaway is both simple and, based on the mounting evidence, increasingly well-supported: feed your gut bacteria well, and they will return the favor.
Sources
- Hidden bugs in our gut appear key to good health, finds global study - University of Cambridge
- Scientists discover a hidden gut bacterium linked to good health - ScienceDaily
- Overlooked group of gut bacteria is key to good health - Medical Xpress
- Healthy adult gut microbiota sustains its own vitamin B12 requirement - Frontiers in Nutrition